In 2001, at a talk by MIT professor of polymer science and engineering Ioannis Yannas, Jordan Miller discovered his vocation. A renowned expert on fibers and polymers at MIT, Yannas had culminated years of work implanting artificial skin on burn patients and was explaining his progress in the field when a young Miller approached him eager to join his research lab. Twenty years later Miller is revolutionizing the field of bioengineering as his breakthrough techniques for bioprinting tissue get him closer to 3D printing replacement organs.
Most people never forget their first encounter with success, and for Miller, learning how to create artificial tissue that functions just like the real thing has been a life-long aspiration. Eager to progress further in the field of bioprinting, Miller began investigating the complexity of organ architecture, particularly, how to provide a blood vessel structure for tissue constructs.
3DPrint.com spoke to Miller to understand his work and what lays ahead for the field of bioprinting, and at that moment, the scientist described that his entire philosophy was based on the concept of vascular architecture for mimicking tissue. He explained how the complexity of the vascular architecture is a question that remains unsolved today and one that he has been focusing on for many years.
“There is an enormous reason to be very excited as the technological capabilities available for research are growing exponentially. We are continually watching the world of cell biology to understand what is happening inside the cells, but everything outside of the cell is our domain expertise—particularly focusing on the extracellular space as we try to solve the decades-old question of how to build vascular architecture. If we could work this out, we are certain that we could move the field of cell biology way further.”
The Role of 3D Printing in Medicine
Along with a team of bioengineers, Miller, now an assistant professor of Bioengineering at Rice University, developed a new bioprinting technique last year called SLATE, short for “stereolithography apparatus for tissue engineering.” It allows scientists to create intricate multi-vascular networks that mimic those found in the body and are an essential passageway to deliver blood, oxygen, and nutrients to organs and tissue.
The breakthrough open-source bioprinting technology, which uses additive manufacturing to make soft hydrogels, was deployed for a proof-of-principle model of a lung-mimicking air sac that contains blood vessel networks 300 microns (0.3 millimeters) in diameter and do not touch neighboring airways. This model is useful for testing how to deliver oxygen to flowing human red blood cells. Additionally, Miller’s team reported experiments to implant bioprinted constructs containing liver cells into mice.
“There is nothing in the known universe as complicated as the structures inside our bodies, so this is a really amazing manufacturing challenge and an incredibly exciting scientific opportunity to investigate,” said Miller. “It’s hard enough to recreate one vessel network with cell-compatible material, and we know that everyone struggles with this. But our organs don’t have just one vascular network, they have lots of vascular architecture. With SLATE, we realized that the capability of our new technique gave us unprecedented design freedom so that we could really address multivasculature.”
Biocompatible Materials and Processes
Like others in the field of bioengineering, he is focused on creating vasculature that can make artificial organs function. Miller saw a great opportunity to utilize the field of 3D bioprinting to address fundamental questions in biology, considering that there is no better way to make architecture than 3D printing. The technology has been around for almost 40 years, yet the expert is adapting it “to deal with the biological realm in a better way that would give us more complexity of structure, and thereby, more complexity of function.”
“We figured out how to modify biomaterials that have historically been used to make solid pieces of tissue-like material, based on photopolymerization,” Miller said. See also: Best 3D Printer Upgrades That Actually Improve Pri…. “The idea is that we can pour a liquid solution into a customized 3D printer, and then shine a blue light on it to induce a chemical reaction that will crosslink that solution into a water-based material that has the consistency of gelatin to form biocompatible materials. However, there was a limit to our process, so we made modifications to the chemistry that would allow us to address and uncover new design capabilities that were impossible before.”
Clinical Applications and Case Studies
Actually, instead of relying on some of the common toxic, light-blocking chemicals used in stereolithography, like Sudan I, which is toxic and can induce cancer, the team decided to go for synthetic and natural food dyes, like tartrazine (yellow food coloring), to enable the stereolithographic production of hydrogels containing intricate and functional vascular architecture.
As Miller and his team continued to work on developing the necessary tools for bioengineering, their proprietary hardware helped them bioprint cells encased in soft gels that act just like vascular networks.
A huge supporter of open source science, Miller said that all source data from the experiments in the published Science study are freely available. In addition, all 3D-printable files needed to build the stereolithography printing apparatus are available, as are the design files for printing each of the hydrogels used in the study.
Moreover, Miller’s bespoke stereolithography machines follow the RepRap philosophy. He even explained that making customized technology was necessary because no other machine could support the work; emphasizing how innovation has led him to develop a series of different technologies that are all trying to address the same question: how to make blood vessel structures.
Regulatory Considerations and Safety
Replicating one of the main functions of the lung was a breakthrough achievement for Miller, who does most of his research at the Physiologic Systems Engineering and Advanced Materials Laboratory at Rice University, known to most as the “Miller Lab.” In fact, the team was able to create an artificial version of an air sac that has multiple independent vascular structures. Something he says is “very similar to how the lung works where you have the airways getting in extremely close proximity to the bloodstream but without touching.” In fact, if blood were to leak from the blood vessels into the windpipe or airways into the main lung, it would result in pulmonary hemorrhage, which can be fatal.
“When we created an artificial version of an air sac, we were able to show that the oxygen pumped into the air sac could oxygenate human red blood cells flowing through the blood vessels. Even more so, we were able to quantitatively measure that,” Miller said. “Just like doctors use blood gas analyzers to measure oxygen in a blood sample for patients, we used the same machine to measure the efficiency of blood oxygenation, except our patient was a 3D-printed gel. What this really proved was that oxygen was getting out of the air sac, diffusing across the layers of tissue-equivalent material that we had 3D printed, then getting into the blood vessel architecture, to finally be taken up by the red blood cells and the hemoglobin within them.”
Research Breakthroughs and Innovations

Photograph of a printed hydrogel containing the distal lung subunit during RBC perfusion while the air sac was ventilated (Credit: Jordan Miller/Rice University)
As Miller poetically described the architecture of organs, he also delved into the innovative process behind his investigation and how he is pushing the limits of creative imagination at the Miller Lab. Working with a team of four graduate students and seven undergraduate students, mostly from the field of bioengineering, means actively progressing investigations as they deliberately work on the gradual design, methodology, and very strong data required to publish their work.
“The research is about more than just lung tissue. See also: Best Budget 3D Printer Upgrades That Actually Impr…. Our goal is to better understand the link between structure and function in biology. We are trying to make new tissue designs that mimic the form that we see in real organs. The question is still whether they can function accordingly. It is a multidecade question that is still open, but one we are hoping to answer in the next five to 10 years.
The Future of Bioprinting and Medical AM
“In addition to the architecture designs, we are also showing a similar capability to make progress on liver tissue engineering. For that, we made a blood vessel-like structure and injected it with capillary cells. Then, we added liver cells into the tissue, and finally evaluated whether they would survive transplantation in mice with chronic liver injury. As the first step in the right direction, we were able to show that the tissue survived and that the cells were beginning to function after two weeks.”
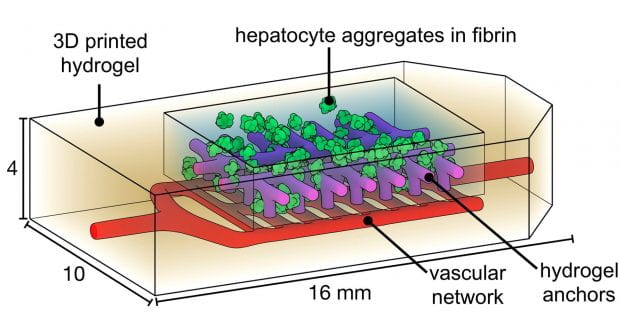
Experiments performed by Rice University and University of Washington researchers explored whether liver cells called hepatocytes would function normally if they were incorporated into a bioprinted implant and surgically implanted in mice for 14 days (Credit: Jordan Miller/Rice University)
This experience was very interesting and complex. Miller suggested that there are several decades of work left to be done in the area, which is still in its infancy.
“Philosophically, it is not clear that we can ever know everything, but we are getting to where we know enough. There are ways to work alongside nature and human biology that is more collaborative than what the terminology of tissue engineering entails. Moreover, we need to create a structure that integrates seamlessly with the human body, that can be remodeled with the human body, and integrates completely. We hope that what we implant in the future will leave no remnant because the body has taken over it and replaced it with native tissue, with cells from the patient. This is the best scenario: to leave no trace.”
The Role of 3D Printing in Medicine
The post Bioengineer Jordan Miller: Clinical Trials for 3D-Printed Organ Replacements Could Start in 5 to 10 Years appeared first on 3DPrint.com | The Voice of 3D Printing / Additive Manufacturing.
from Your daily news from 3DPrint.com https://bit.ly/3di3A1Y
Related: Medical Center Completes Clinical Trials of 3D-Printed Nasal Swabs · 3D Printed Drug Delivery Implant Achieves Successful Clinical Trial for Parkinso · Robot Skin 3D Printer Close to First-in-Human Clinical Trials
Frequently Asked Questions
How is 3D printing used in medicine?
3D printing is used in medicine for surgical planning models, custom implants, bioprinting tissue scaffolds, drug delivery systems, dental aligners, and prosthetics. It enables patient-specific solutions that improve outcomes and reduce surgery time.
What materials are biocompatible for 3D printing?
Common biocompatible materials include PEEK, titanium alloys (Ti6Al4V), bio-ceramics (hydroxyapatite), medical-grade resins, PLA for temporary implants, and hydrogels for bioprinting. Material choice depends on the application and required mechanical properties.
Is 3D printed medical equipment FDA approved?
Yes, several 3D printed medical devices have FDA clearance, including orthopedic implants, dental restorations, and surgical guides. Each device must go through the appropriate regulatory pathway based on its risk classification.
📌 Related Articles
- Best 3D Printer Upgrades That Actually Improve Print Quality: Complete 2026 Guide
- Best Budget 3D Printer Upgrades That Actually Improve Print Quality: Belts, Springs, Hotends & More
- 3D Printing News: Multi-Material Breakthrough and Aerospace Market Boom
- 3D Printing Tungsten Carbide: How Hot-Wire Laser Technology Creates Metal Harder Than Steel
- ABS 3D Printing Settings Guide: Temperature, Enclosure, and Cooling for Strong Parts


