The COVID-19 pandemic has changed the world as we once knew it in many ways, beginning with renewed value for good health and not sweating the small stuff; however, for those who have been affected by coronavirus, the story is much different. Known to be a frightening experience even for individuals who may be considered to be in good shape, this potentially life-threatening virus often means hospitalization. As beds in hotspot areas like New York have filled to capacity, the shortage for personal protection equipment (PPE) has been well-known, and widespread.
 While medical users and the open-source community around the world have been extremely resourceful in 3D printing much-needed medical devices like ventilators, companies like Hybrid Manufacturing Technologies have stepped up to produce the volumes needed in the US for PPEs. The Mckinney, Texas-headquartered manufacturer has announced development of a new technique called AXIOM, combining 3D printing with injection molding.
While medical users and the open-source community around the world have been extremely resourceful in 3D printing much-needed medical devices like ventilators, companies like Hybrid Manufacturing Technologies have stepped up to produce the volumes needed in the US for PPEs. The Mckinney, Texas-headquartered manufacturer has announced development of a new technique called AXIOM, combining 3D printing with injection molding.
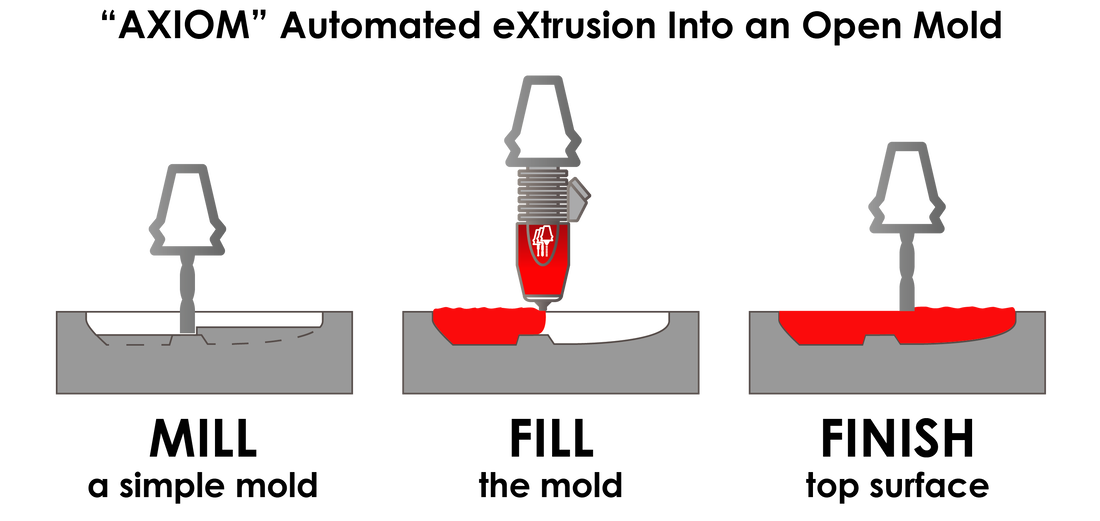
The benefits of 3D printing continue to be discovered—and expanded upon—within the medical realm. Sometimes, though, the combination of traditional and progressive technologies can be extremely effective for making necessary components. The AXIOM (Automated eXtrusion Into an Open Mold) process is accompanied by the AMBIT XTRUDE head in a milling machine.
XTRUDE head in a milling machine.
Although 3D printing enthusiasts currently enjoy advantages such as affordability and accessibility in hardware, software, and materials, injection molding is still often the first choice for users seeking better surface finish and quality in parts. 3D printing may also be beneficial for low-volume production, but fabrication of large quantities may not always be realistic—especially in comparison to molding.
The Role of 3D Printing in Medicine
“These factors have been highlighted in recent efforts to address PPE shortages arising with the COVID-19 pandemic,” stated the Hybrid Manufacturing Technologies team in a recent press release.
Biocompatible Materials and Processes
The AXIOM process works in three steps: mill, fill, and finish. This allows for plastic parts like face shields to be fabricated quickly during sudden surges and peak demand from the medical community, especially during a viral pandemic. It takes less than one minute to produce a polypropylene part, with smooth surfaces for easy sterilization—a critical requirement in the medical setting.
“We have brought together 3D printing, machining, and molding in a new way to supplement supply chains as demand surges. This means producing polymer parts without layers or the surface roughness associated with 3D printing at speeds approaching injection molding.” said Dr. Jason Jones, CEO, Hybrid Manufacturing Technologies.
Clinical Applications and Case Studies
These parts have not been tested for any regulatory compliance. As such, use of this design and/or process is done at your own risk.
What do you think of this news? Let us know your thoughts! Join the discussion of this and other 3D printing topics at 3DPrintBoard.com.
[Source / Images: ‘Hybrid Manufacturing Technologies’]
Regulatory Considerations and Safety
The post AXIOM Process Combines 3D Printing with Injection Molding, Prompted by COVID-19 Needs appeared first on 3DPrint.com | The Voice of 3D Printing / Additive Manufacturing.
from Your daily news from 3DPrint.com https://bit.ly/2AgK7RM
Frequently Asked Questions
How is 3D printing used in medicine?
3D printing is used in medicine for surgical planning models, custom implants, bioprinting tissue scaffolds, drug delivery systems, dental aligners, and prosthetics. It enables patient-specific solutions that improve outcomes and reduce surgery time.
What materials are biocompatible for 3D printing?
Common biocompatible materials include PEEK, titanium alloys (Ti6Al4V), bio-ceramics (hydroxyapatite), medical-grade resins, PLA for temporary implants, and hydrogels for bioprinting. Material choice depends on the application and required mechanical properties.
Is 3D printed medical equipment FDA approved?
Yes, several 3D printed medical devices have FDA clearance, including orthopedic implants, dental restorations, and surgical guides. Each device must go through the appropriate regulatory pathway based on its risk classification.
📌 Related Articles
- The Current State of Metal 3D Printing in 2020
- How Divergent Technologies Is 3D Printing Cruise Missiles for the U.S. Military
- ABS 3D Printing Settings Guide: Temperature, Enclosure, and Cooling for Strong Parts
- 3D Printing Safety Equipment Guide: Respirators, Gloves, and Ventilation for 2026
- Bambu Lab P1S Review: The Enclosed CoreXY Speed Demon That Just Works