US researchers have been working to improve methods for detecting carbon monoxide, releasing the details of their recent study in ‘Additive Manufacturing of a Flexible Carbon Monoxide Sensor Based on a SnO2‐Graphene Nanoink.’
Hundreds of people are killed each year due to accidental carbon monoxide poisoning. Because the gas is odorless and has the potential for building up quickly in a home or other structure, the danger to humans is very real. While there are numerous sensing units on the market today relying on the semiconductor SnO2, the researchers created a new process for fabricating sensor arrays founded on ‘semiconductor catalyst‐decorated reduced graphene oxide sensor media.’
Current manufacturing for sensors is expensive, time-consuming, and requires very specific conditions such as high temperatures and numerous steps in production:
“In addition, the bulk SnO2 sensor media typically must be heated to temperatures above 400°C to achieve a sufficient CO response,” stated the researchers. “This temperature limit hinders the development of polymer film‐supported flexible and thin devices.”
Why Calibration Matters
Two different methods have been used to improve manufacturing of sensors, using nano‐sized SnO2 that could be dispersed in fluids or thin films with an SnO2 base, created via solution processing. While there has been some success in these previous methods, the authors point out that high-temperatures are still required, or a heater was needed for activating a response. Other research has reported low-temperature sensing devices, but processes were found to be “neither scalable or of high technological viability.”
For this study, the researchers created a nanoink made up of high response SnO2‐decorated rGO. The result was a “fully printed, flexible sensor” able to detect CO at room temperature.
“The inks used to fabricate the devices studied here were produced by the re‐dispersion of the purified SnO2‐rGO aerogel. To improve the dispersion quality and stability, the aerogel was firstly ground using a mortar and pestle and then different solvents were tested, including NMP, ethylene glycol, and ethanol. In several cases, sonication was performed for 10–12 h to obtain a good and stable dispersion,” stated the researchers. “The final ink had its surface tension and viscosity characterized using, respectively, a DSA100 tensiometer from Kruss and a DV3T rheometer from Brookfield.”
Step-by-Step Calibration Process
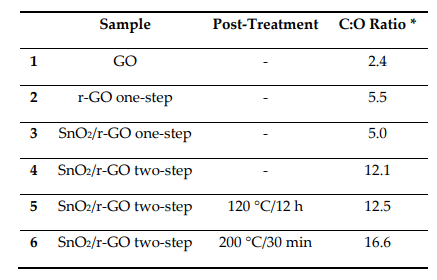
C:O atomic ratio calculated from the XPS spectra obtained for the drop‐cast films from NMP dispersions. All the samples were dried in an oven at 70 °C for 30 min and some of them were then submitted to the extra post‐treatment step, as indicated. GO = commercial graphene oxide; r‐GO = reduced graphene oxide, submitted to the solvothermal process for reduction; SnO2/r‐GO = SnO2 decorated reduced graphene oxide. * C:O ratio for SnO2‐containing samples was calculated by subtracting the oxygen atoms linked to Sn.
This new technique offered better results due to the loading of SnO2, dispersion, and nucleation on graphene sheets. See also: Best 3D Printer Upgrades That Actually Improve Pri…. Films were then annealed further, with the reduction of graphene oxides leading to the success in the room temperature environment. Conductivity was also increased where films were annealed, meaning that the researchers were able to use thinner films and enjoy better gas response.
Fine-Tuning Print Quality
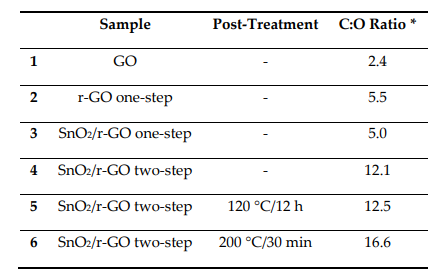
X‐ray diffraction data for the SnO2‐rGO composite synthesized in this work.
Ultimately, the researchers noted that inkjet was suitable for printing, allowing them to create flexible metal traces in high resolution (with commercially available nano-ink). This technique was also used for the fabrication of the devices’ active layers.
Troubleshooting Common Issues
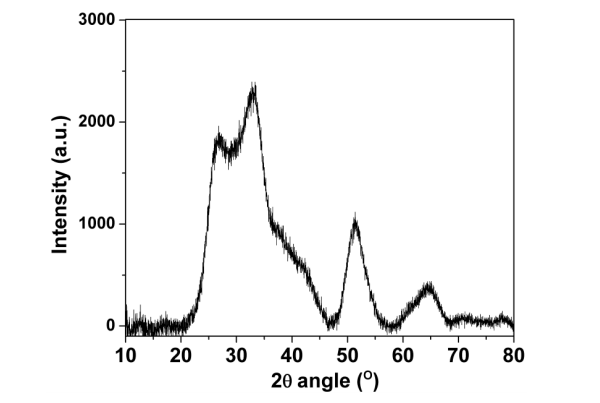
High‐resolution XPS scans for the selected SnO2‐graphene peaks assigned to (A) carbon; (B) oxygen; (C) tin. Peaks were fitted and deconvoluted using Hawk analysis software.
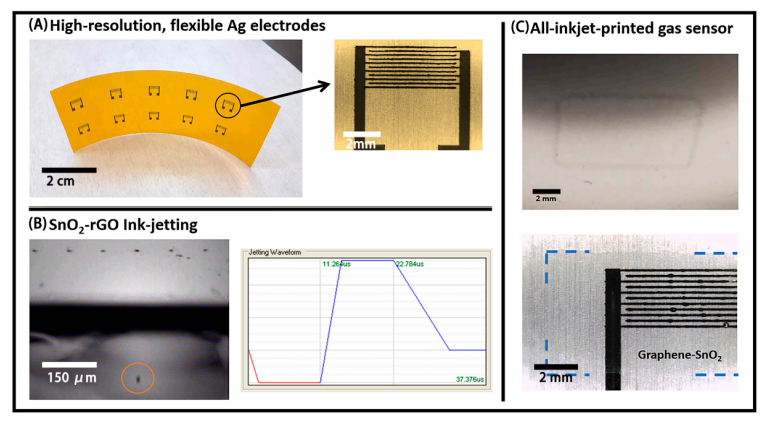
(A) Inkjet‐printed Ag Interdigitated electrode structures (IDEs) arrays on Kapton; (B) successful jetting of a single droplet of the SnO2‐rGO dispersion in NMP (left) and the respective waveform used for that (right); (C) 1‐layer inkjet‐printed film of SnO2‐rGO on bare poly(ethylene theraphthalate) (PET) (top) and 2‐layers inkjet film of SnO2‐rGO on Ag IDEs (bottom). Blue traced lines are included to guide the eyes on the last picture, due to the low contrast in the optical microscopy image.
“The specificity of the all‐printed sensor to CO was demonstrated primarily through a control measurement in which the device was tested in presence of H2. Upon H2 exposure, a much smaller variation of resistance (ca. 4%) was recorded, demonstrating the higher reactivity of CO molecules towards SnO2. A test for CO with a sensor having only rGO in the active layer (no SnO2) showed sensitivities of only 2−4% (results not shown).
Maintaining Consistent Results
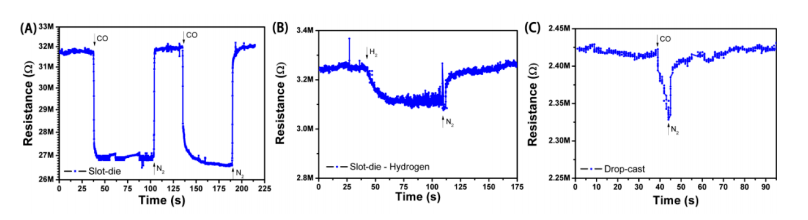
Detection curves for: (A) slot‐die coated film of SnO2‐rGO against CO; (B) slot‐die coated film of SnO2‐rGO against H2; (C) drop‐cast film of SnO2‐rGO against CO.
“The sensors reported here operate at room‐temperature, are thin and flexible, and can be additively fabricated through scalable, cost‐effective protocols that can enable innovative applications and their integration into different products.”
3D printing is becoming increasingly popular for the fabrication of a variety of sensors, from tactile sensors to those that are embedded for monitoring purposes, made with conductive materials, and more. What do you think of this news? Let us know your thoughts! Join the discussion of this and other 3D printing topics at 3DPrintBoard.com.
Why Calibration Matters
[Source / Images: ‘Additive Manufacturing of a Flexible Carbon Monoxide Sensor Based on a SnO2‐Graphene Nanoink’]
The post 3D Printing Carbon Monoxide Sensors with SnO2 Graphene Nanoink appeared first on 3DPrint.com | The Voice of 3D Printing / Additive Manufacturing.
Step-by-Step Calibration Process
from Your daily news from 3DPrint.com https://bit.ly/2MF0o5R
Related Articles
Frequently Asked Questions
Why is 3D printer calibration important?
Calibration ensures dimensional accuracy, proper layer adhesion, and consistent print quality. Without it, parts may have fitting issues, poor surface finish, or structural weaknesses. Regular calibration compensates for mechanical wear and environmental changes.
How often should I calibrate my 3D printer?
Calibrate after any hardware changes, when switching filaments, or if you notice quality degradation. A basic bed level check before each print is good practice, while full calibration (e-steps, flow rate, PID tuning) should be done monthly or after maintenance.
📌 Related Articles
- Best 3D Printer Upgrades That Actually Improve Print Quality: Complete 2026 Guide
- Bambu Lab A1 vs Elegoo Centauri Carbon: Full Specs Comparison & Buyer’s Guide
- Best Budget 3D Printer Upgrades That Actually Improve Print Quality: Belts, Springs, Hotends & More
- Prusa Research Mini+ vs Prusa MK4: Full Specs Comparison & Buyer’s Guide
- ABS 3D Printing Settings Guide: Temperature, Enclosure, and Cooling for Strong Parts