The American Society of Mechanical Engineers (ASME) hosted a first-of-its-kind event with experts discussing the instrumental role and impact of additive manufacturing (AM) on patient care. Originally set to take place at the Minneapolis Convention Center, the AM Medical: Additive Manufacturing & 3D Innovations summit went virtual on May 27 due to the ongoing COVID-19 pandemic. The interactive experience provided a forum for clinicians, engineers, and manufacturers to discuss the role of 3D printing and bioprinting to advance personalized health care.
Featuring over a dozen talks and panels with regional, national, and international leaders, the AM Medical event focused on the setbacks, regulations and successful experiences of some of the world-leading companies and healthcare institutions that need to address new issues in medical additive manufacturing on a daily basis as they seek to democratize the technology.
During the summit’s welcome session, ASME’s executive director and CEO, Thomas Costabile, said that “with over three million patients directly impacted through 3D printing, it won’t be long before everyone knows someone who has personally experienced the power of the technology to transform lives.” Costabile also described how advances in 3D printing applications enable better and quicker devices to market and for patient care. Highlighting that improving point-of-care in hospitals with 3D printing has brought together engineers and physicians that are working together to develop devices for the future of healthcare. “It’s a better quality of life for all of us,” he concluded.
The Role of 3D Printing in Medicine
Participants at the event had an opportunity to hear about advancements in 3D printing, materials, quality processes, scanning, visualization, regulations, along with new software solutions for post-processing, modeling, and simulation. Attendees were able to choose from over 30 technical sessions across four tracks: medical device manufacturing, dental applications, biofabrication, and point-of-care manufacturing and clinical applications.
During one of the first sessions of the day, community leaders from the United States Veterans Affairs (VA) Puget Sound Healthcare Center, the Montefiore Medical Center in New York, and Johnson & Johnson joined in to share some of the efforts, resources, and needs of healthcare professionals today as they step up to deal with the short-term healthcare consequences of the current pandemic.
Set to transform the medical AM community, the current response to COVID-19 could reshape healthcare institutions in the long-run. The pandemic has disrupted the response of healthcare providers, drawing attention and resources to the AM industry, and spurring the use of 3D printing in hospitals, manufacturing facilities with 3D printing capabilities and designers that are collaborating to ensure the delivery of safe personal protective equipment (PPE) for medical caregivers in the front-lines.
“There is a lot of additive manufacturing power across this country. Having all of that manufacturing power is a huge help because we can rapidly prototype designs to get around supply chain deficiencies,” said Beth Ripley, director of the Veterans Health Administration (VHA) 3D Printing Network, and a panelist in the Medical AM/3DP Community Response to COVID-19 presentation. “Thinking about how close this has brought manufacturing and hospitals together is a real win for all of us. We have seen some great collaborations and help from engineering and industry partners and that’s going to drive forward how we practice within the hospitals post-pandemic.”
Biocompatible Materials and Processes

Beth Ripley performs a quality check on a 3D printed model of a kidney with a cancerous tumor (Credits: Chris Pacheco/VA)
As sessions overlapped to cover the entire program in only eight hours, attendees were encouraged to participate in Lunch & Learn sessions with experts from Protolabs, B9Creations, and ZEISS. From virtually anywhere, participants had the chance to meet peers and speakers for a unique networking opportunity, as well as visit the sponsors and exhibitors hall which included stands by industry leaders like 3D Systems, CAD BLU, EOS, Formlabs, GE Additive, Marvel Medtech, MasterGraphics, NCS Technologies, Nota3D, Protolabs, Trumpf, and Xometry.

Clinical Applications and Case Studies
Titanium femoral implants additively manufactured by GE Additive (Credits: GE)
After lunch, keynote speakers from the Mayo Clinic, Stryker, Advanced Regenerative Manufacturing Institute (ARMI)/BioFabUSA, and the Food and Drug Administration (FDA) convened to discuss effective quality systems for additive manufacturing processes that guarantee that medical products are safe and effective for use.
“The FDA is really looking at the end product, so whether you are using AM or conventional machining, all of the quality systems requirements at the high level are still the same. Things change when you get into the nitty gritty of controlling, so we are trying to make sure that our investigators have the right knowledge needed to appropriately evaluate quality systems at these AM locations,” suggested Matthew DiPrima, a materials scientist at the FDA, as he delved into how the agency interacts with companies to make sure that products safely make it out to American patients.
Regulatory Considerations and Safety
With so many panels discussing the importance and uncertainty surrounding the regulation of 3D-printed medical devices, DiPrima’s talk was a big focal point of the day. The expert reassured everyone that the FDA had “invested a lot of time and money into keeping up to date with the technology,” claiming that, in only six years, its footprint had expanded significantly to stay on top of the technology and any changes.
“The next big question for us will be point-of-care, [from] spending time trying to understand quality systems for device production at a hospital [to] collaborating with the DoD [Department Of Defense] hospital systems on test cases. We are doing our best to stay at the edge of technology, but at the end of the day, we still have to react to what the industry is doing,” he concluded.
Research Breakthroughs and Innovations
Another highly anticipated meeting brought together experts in the field of biofabrication, to talk about the needs of bioprinted organs, particularly the tools and infrastructure that will be needed for storage, transportation, surgeries, and other challenges that will have to be addressed to ensure healthcare professionals will be able to effectively use the bioprinted tissues. Renown Turkish scientist and academic at Pennsylvania State University, İbrahim Özbolat, along with CSO and founder of Embody, Michael Francis, and the principal scientist of biologics at Medtronic, Bre Jacobs, explained some of the biggest challenges that lie ahead.
“Hopefully, in the next 10 to 15 years we will create tissue-like structure at the organ level […] making functional tissue that is fully vascularized and anatomically like human organs for transplant with minimal post-transplantation problems,” declared Özbolat.
The scientist suggested that a major issue ahead of bioprinted organs will be the cold chain management for transporting human-derived stem cell packages from patients to organ manufacturing facilities. He also considered that, when it comes to the use of a patient’s primary cells, organ manufacturing facilities should be developed close to the hospitals to allow for cells to be isolated, expanded, printed, and implanted back into the patient, without placing the cells at risk. Francis also examined future demands in biofabrication, suggesting that there is an opportunity to advance the field of transportation of biomanufactured tissues as one of the big next forefronts in bioprinting: “A lot of our needs are unique and we are going to have big breakthroughs.”
“Although there are a lot of companies entering the space that are helping supply clinical-grade feedstock and raw materials for cell printing, the biggest hurdle I see is probably the cost to market […] which is tremendously expensive,” indicated Francis.
As the event came to an end, one of the final panels summed up the future of medical and dental 3D printing, with Ripley returning as a keynote speaker alongside Sam Onukuri, from Johnson & Johnson; Medtronic’s Markus Reiterer, and Adam Jakus, co-founder and CTO of Dimension Inx, who described what could come next for medical 3D printing: “likely, the first bioprinted product with live cells entering the market will be tissue-engineered for the musculoskeletal space.”
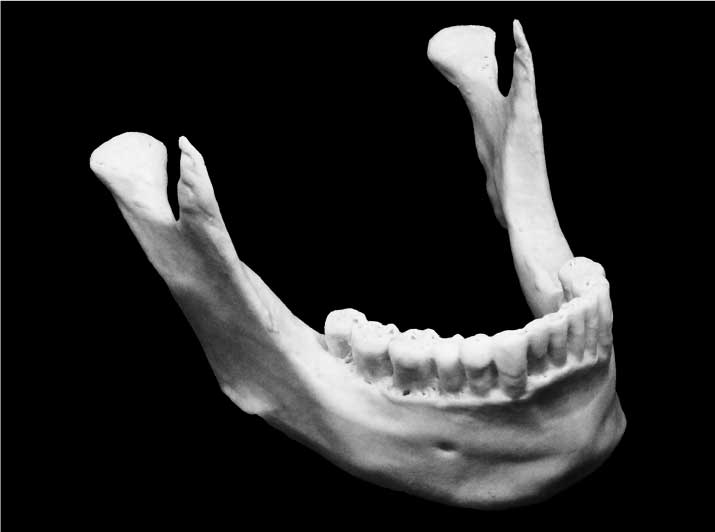
Biocompatible Materials and Processes
Patient-matched mandible 3D printed with Hyperelastic Bone® by Dimension Inx. (Credits: Dimension Inx)
With so many experts discussing critical aspects of the advantages and limitations of additive manufacturing applied to the medical industry, like how to deliver customized products, or how the medical additive manufacturing community is using the technology to make anatomical models and implants, even how to implement an integrated 3D printing strategy to evolve into viable production method for end-use medical parts, it was hard to choose which session to listen in. Luckily for viewers, ASME will be posting recordings of the sessions on its AM Medical site.
Even though, early on during the seminar, event managers from ASME had to deal with a few technical glitches, those were eventually fixed and audiences could tune into the sessions and workshops. Hosting the conference virtually and free was a great way for the organization to tap into a wider audience around the world, especially as interest in AM in the medical industry continues to open the market to new companies with disruptive ideas that could translate research from the lab straight to the patient. After all, that is what the medical industry needs to assure, patient care, and 3D printing is helping to fulfill what other traditional technologies could not.
Clinical Applications and Case Studies
The post Virtual AM Medical Event: From Innovations to the Future of Additive Manufacturing in the Medical Industry appeared first on 3DPrint.com | The Voice of 3D Printing / Additive Manufacturing.
from Your daily news from 3DPrint.com https://bit.ly/3ewqcx0
Related: AMFG Publishes Additive Manufacturing Landscape Report 2020: Breaking Down the I · Here’s How Much Medical Equipment the World’s Additive Manufacturing Equipment C · AMS 2020: Panels on 3D Printing in Implants and Orthopedics, Regulation in Addit
Frequently Asked Questions
How is 3D printing used in medicine?
3D printing is used in medicine for surgical planning models, custom implants, bioprinting tissue scaffolds, drug delivery systems, dental aligners, and prosthetics. It enables patient-specific solutions that improve outcomes and reduce surgery time.
What materials are biocompatible for 3D printing?
Common biocompatible materials include PEEK, titanium alloys (Ti6Al4V), bio-ceramics (hydroxyapatite), medical-grade resins, PLA for temporary implants, and hydrogels for bioprinting. Material choice depends on the application and required mechanical properties.
Is 3D printed medical equipment FDA approved?
Yes, several 3D printed medical devices have FDA clearance, including orthopedic implants, dental restorations, and surgical guides. Each device must go through the appropriate regulatory pathway based on its risk classification.
📌 Related Articles
- The Current State of Metal 3D Printing in 2020
- 3D Printing News: Multi-Material Breakthrough and Aerospace Market Boom
- Best 3D Printer Upgrades That Actually Improve Print Quality: Complete 2026 Guide
- ABS 3D Printing Settings Guide: Temperature, Enclosure, and Cooling for Strong Parts
- 3D Printing Safety Equipment Guide: Respirators, Gloves, and Ventilation for 2026



