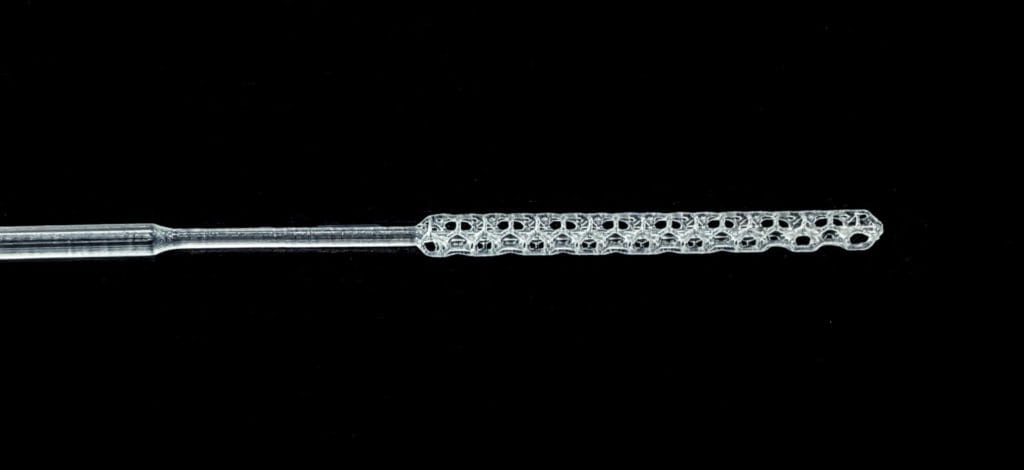
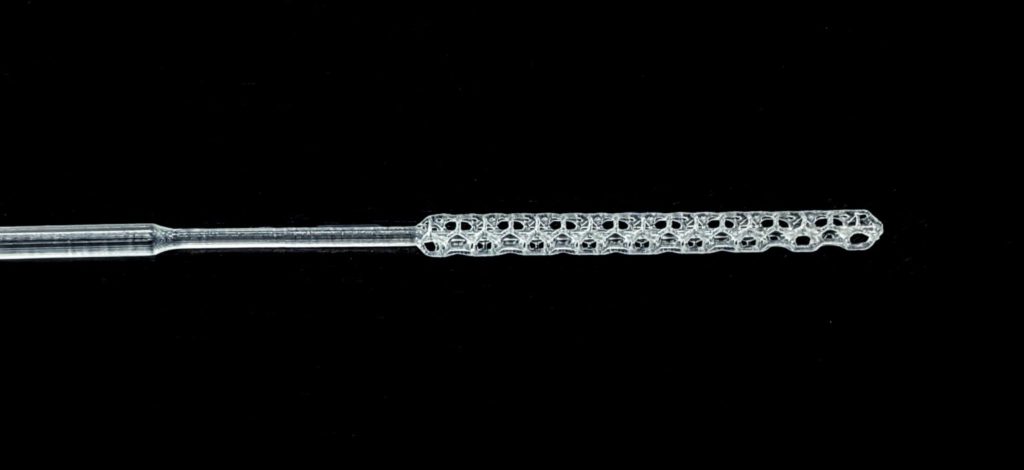
A year ago, if you would have asked anyone in our industry what they thought might be a future killer application, it’s highly doubtful that anyone would have replied with, “nasopharyngeal swabs.” Until recently, it was a niche product and the entire market was serviced by a few dominant industry players.
While there are now other protocols, the main test for COVID-19 testing involves gathering virus from deep in a person’s nasal cavity. The SARS-CoV-2 virus is collected using a nasopharyngeal (NP) swab. Traditionally, these swabs are made in two parts, including a polyester handle and a tip with tiny rayon fibers called flock.
The two primary companies that make them, Purtian Medical Products Co. and Copan Diagnostics, bulk manufacture them in multiple steps, which include production, assembly, sterilization and packaging, among others. Their process requires customized machinery and a sizable group of relatively skilled people.
When the global pandemic struck, the demand for COVID-19 test kits skyrocketed, far outpacing the combined capacity of these two companies. For several reasons, they had difficulties scaling their businesses. They both produce many other products for the medical industry and adding new equipment is a timely endeavor. To make matters worse, Copan which is located in Italy (a hotspot for the virus) was challenged with maintaining the health of its own workforce.
3D Printing to the Rescue
As it became apparent that the normal suppliers couldn’t fully meet the need, the additive manufacturing industry began working on the problem. Markforged, a manufacturer of filament-based 3D printers, partnered with Neurophotometrics to produce 3D-printed NP swabs made from their Fiberflex Rayon.
Separately, Northwell Health teamed up with the University of South Florida, Tampa General Hospital and Massachusetts-based Formlabs and worked with physicians to design their own NP swab, which Formlabs recently started printing in its FDA-registered, ISO 13485-certified factory in Ohio.
Results from a clinical trial of 3D-printed NP swabs. Image courtesy of Beth Israel Deaconess Medical Center.
Soon after, a consortium of 3D printing companies was codified. Their goal was to deliver clinically tested, FDA-registered, 3D-printed COVID-19 NP test swab designs with superior or equivalent efficacy to flocked swabs, at scale.
Origin Partners with Henkel
Origin, manufacturer of stereolithography (SLA) 3D printers was one of the founding members of the consortium. It began working with several partners to develop what it is now calling the world’s first FDA-compliant, sterile, 3D-printed NP test swab.
In a new announcement, the startup is providing more detail about the process. Origin collaborated with materials company, Henkel and Beth Israel Deaconess Medical Center (BIDMC) on the swab’s development. Working with generative design software, Origin was able to design a one-piece swab that performed as well as the traditional version. Henkel leveraged its own Albert software platform to specify a material that would meet the requirements for an in-body medical device. Together they tested the design’s clinical capabilities, in addition to validating each step in the sterilizations process, and conducting rigorous mechanical testing and packaging certification.
Scalability and Speed-to-Market
Within weeks they were able to bring a product to market that is classified as a sterile device and is considered a finished medical product, which is regulated by the FDA.
It’s a perfect example of two of 3D printing’s biggest benefits: scalability and speed-to-market. As Chris Prucha, Founder and CEO at Origin noted in the press release, “by working collaboratively and utilizing each other’s technologies, we identified, optimized and scaled the manufacturing process to bring an application to market extremely fast.”
Origin’s sterile NP swabs are currently shipping to leading healthcare facilities, government institutions, and independent testing centers in the U.S. See also: Best Budget 3D Printer Upgrades That Actually Impr…. and several other countries. They’re also available for purchase on Amazon.
But beyond the opportunity with NP swabs, this collaboration also further substantiates the industry’s growth into functional part production.
In the press release, Ken Kisner, Head of Innovation for 3D Printing at Henkel said, “From inception, the vision behind Henkel’s Open Materials Platform was to enable collaboration all along additive manufacturing’s value chain. Working together with Origin, we were able to develop a product which is just as effective as its mass-produced counterpart. With the constraints commercial medical suppliers are facing, this presents a significant opportunity for the 3D printing industry to demonstrate its capabilities, beyond prototyping.”
Innovate Globally, Produce Locally
The problem wasn’t just related to the manufacturing of NP swabs. There were constraints all across the medical supply chain. Some of it had to do with the traditional model of centralized manufacturing and logistics. The healthcare industry relies on a relatively small number of producers and distributors. When they’re impeded, all bets are off. Further, the vast number of products, the niche nature of some of them, and shelf life issues make some medical products difficult to stockpile.
Perhaps more than anything else, this application demonstrates the value of a nimble, distributed manufacturing network, where identical parts can be made as close as possible to the point of need. In some cases, it can be financially beneficial, but in others like this decentralizing production provides an insurance policy in the event the unimaginable happens. We know it can, because it has.
The post NP Swabs Prove 3D Printing’s Scalability and Speed-to-Market Advantages appeared first on 3DPrint.com | The Voice of 3D Printing / Additive Manufacturing.
from Your daily news from 3DPrint.com https://bit.ly/2N2ffHA
Frequently Asked Questions
How is 3D printing used in medicine?
3D printing is used in medicine for surgical planning models, custom implants, bioprinting tissue scaffolds, drug delivery systems, dental aligners, and prosthetics. It enables patient-specific solutions that improve outcomes and reduce surgery time.
What materials are biocompatible for 3D printing?
Common biocompatible materials include PEEK, titanium alloys (Ti6Al4V), bio-ceramics (hydroxyapatite), medical-grade resins, PLA for temporary implants, and hydrogels for bioprinting. Material choice depends on the application and required mechanical properties.
Is 3D printed medical equipment FDA approved?
Yes, several 3D printed medical devices have FDA clearance, including orthopedic implants, dental restorations, and surgical guides. Each device must go through the appropriate regulatory pathway based on its risk classification.
📌 Related Articles
- The Current State of Metal 3D Printing in 2020
- Best Budget 3D Printer Upgrades That Actually Improve Print Quality: Belts, Springs, Hotends & More
- 3D Printing News: Multi-Material Breakthrough and Aerospace Market Boom
- Best 3D Printer Upgrades That Actually Improve Print Quality: Complete 2026 Guide
- ABS 3D Printing Settings Guide: Temperature, Enclosure, and Cooling for Strong Parts