Quick Answer: What is NeuroPrint Technology?
NeuroPrint is a hybrid 3D printing technology developed by researchers at Saint-Petersburg State University that enables the rapid production of patient-specific neural and muscular implants. Using a RegenHU 3DDiscovery bioprinter, the technology combines ink-jet dispensing of conductive inks with extrusion of silicone insulators and cold-air plasma activation. Key benefits include:
- 24-hour production time from design to prototype (vs. weeks for traditional methods)
- Customizable electrode configurations for spinal cord, brain, or muscle implantation
- Soft, biocompatible materials that integrate with neural tissue long-term
- Cost-effective prototyping accelerating research in neuroprosthetics
This innovation opens new possibilities for treating neurological conditions including spinal cord injuries, epilepsy, Alzheimer’s, Parkinson’s disease, and multiple sclerosis through personalized neural interfaces.
Researchers have developed a new 3D printing technology that makes it possible to rapidly customize muscular and neural implants for monitoring and restoring motor and autonomic functions. The neural interfaces can be used to link brains to computers and could enable customized bioelectronics to treat patients with nervous system problems, like multiple sclerosis, epilepsy, Alzheimer’s, and Parkinson’s disease.
In a new study published in the journal Nature Biomedical Engineering, a team led by Ivan Minev, professor of Intelligent Healthcare Technologies at the University of Sheffield, England, and Pavel Musienko, head of the Neuroprosthetics Lab at Saint-Petersburg State University, in Russia, created a neural implant that was used to stimulate the spinal cord of animal models with spinal cord injuries. The technology now has the potential to develop new treatments for human patients with paralysis.
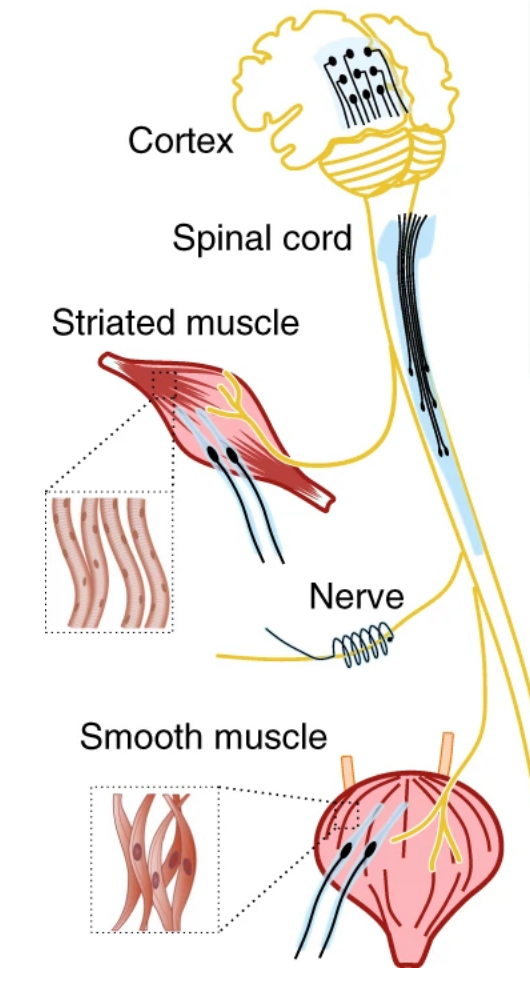
Electrode implants printed from platinum microparticles and silicone can be adapted to the anatomy of electrogenic tissues. Image courtesy of Saint-Petersburg State University/University of Sheffield.
How NeuroPrint Technology Works
This new patient-specific approach is possible due to NeuroPrint hybrid 3D printing technology developed at Saint-Petersburg State University. Using a 3DDiscovery bioprinter from Swiss 3D biotechnology company RegenHU, they created the geometry of the future implant made of silicone – which also serves as an insulating material. Then microparticles of platinum or another electrically conductive element of the implant are applied to the framework, and then the surface is activated by cold plasma.
The integrated platform for hybrid printing combines three key processes:
- Ink-jet dispensing of low-viscosity conductive inks (platinum microparticles)
- Extrusion of insulating silicone pastes for structural support
- In situ surface activation via cold-air plasma treatment
The number and configuration of electrodes in the neural implant can be changed, producing devices for implantation in the tissue of the spinal cord, brain, or muscles. Furthermore, the average production time from project creation to prototyping can take just 24 hours. The capacity of hybrid printing to integrate soft materials and composites into the bioelectronic devices proved well adapted to various anatomical structures and experimental models to investigate, enable, and recover functions of the neuromuscular system.
Research Results and Clinical Potential
Neuroscientists have already exploited the NeuroPrint technology to carry out research on various model objects. Through the monitoring and activation of neuronal pathways in the brain, spinal cord, and neuromuscular system of cats, rats, and zebrafish, the team showed that the printed bioelectronic interfaces permitted long-term integration and functional stability.
"We have tested our development in experiments on freely moving rats for chronic recording of the electrocortical signals of the cerebral cortex, that is a necessary element of the brain-computer interface," said Musienko. "The experiments on paralysed animals have shown that electrical stimulation of neural networks effectively restores locomotor function. Thus, the NeuroPrint technology opens up new opportunities both for basic research into the central nervous system and for neuroprosthetics when people suffer from various diseases and injuries."
According to the research, neuromuscular interfaces are required to translate bioelectronic technologies for application in clinical medicine. Neural interfaces establish communication between biological systems and electronic devices, which is why the possibility to interface with neural circuits has attracted a new generation of researchers and companies, like Neuralink, Elon Musk’s neural engineering company founded to make brain-machine interfaces to help individuals with paralysis. Scientific tool developer Qrons also announced research to develop innovative 3D printable biocompatible materials to treat penetrating brain injuries. While earlier this year, a team of researchers at MIT made implants that are not only soft enough for the human body but conductive enough to interact with the human brain.

Prototype soft bioelectronic implants for use as neuromuscular interfaces. Image courtesy of the University of Sheffield.
Comparison: NeuroPrint vs Traditional Neural Implant Manufacturing
| Feature | NeuroPrint (3D Printing) | Traditional Manufacturing |
|---|---|---|
| Production Time | ~24 hours | 2-8 weeks |
| Customization | Fully patient-specific, easy to modify | Limited to standard designs, expensive to customize |
| Material Flexibility | Soft, biocompatible composites (silicone + metals) | Often rigid metals (platinum, iridium) |
| Cost per Prototype | Low (~$500-1000) | High (~$5000-15000) |
| Electrode Configuration | Customizable number and placement | Fixed arrays |
| Long-term Integration | Proven stable in animal studies | Variable, often causes scarring |
Comparison: Leading Neural Interface Technologies
| Technology | Company/Institution | Key Innovation | Status |
|---|---|---|---|
| NeuroPrint | Saint-Petersburg State University / University of Sheffield | Hybrid 3D printing for patient-specific soft implants | Animal testing complete, clinical translation planned |
| Neuralink | Neuralink (Elon Musk) | High-bandwidth brain-machine interface with robotic insertion | Human trials begun (2024) |
| Utah Array | University of Utah / Blackrock Neurotech | Silicon microelectrode array with 100 channels | FDA-cleared, clinical use since 2000s |
| BrainGate | Brown University / multiple partners | Wireless neural prosthetics for paralyzed patients | Clinical trials ongoing |
| Soft Implants | MIT | Rubber-like materials that match brain stiffness | Animal testing, early research stage |
The Future of Personalized Neural Medicine
Linking the human brain to a computer via a neural interface is an ambition for many researchers. However, innovation in the field is hampered by the huge costs and long development time it takes to produce prototypes, which are needed for exploring new treatments. According to the University of Sheffield, the technology promises great potential to bring new medical treatments for injuries to the nervous system based on a fusion of biology and electronics. The vision relies on implants that can sense and supply tiny electrical impulses in the brain and the nervous system.
Through the new study, the team has shown how 3D printing technology can be used to make prototype implants much quicker and more cost-effectively in order to speed up research and development in the area. The team claims the implants can be easily adapted to target specific areas or problems within the nervous system.
Moreover, using the new technique, a neuroscientist could order a design which the engineering team can transform into a computer model that feeds instructions to the printer. The printer would then apply a palette of biocompatible, mechanically soft materials to realize the design. The implant can be quickly amended if changes are required, giving neuroscientists a quicker and cheaper way to test their ideas for potential treatments.
"The research we have started at TU Dresden (Dresden University of Technology) and continuing here at Sheffield has demonstrated how 3D printing can be harnessed to produce prototype implants at a speed and cost that hasn’t been done before, all whilst maintaining the standards needed to develop a useful device," explained Minev. "The power of 3D printing means the prototype implants can be quickly changed and reproduced again as needed to help drive forward research and innovation in neural interfaces."

The integrated platform for hybrid printing combines ink-jet dispensing of low-viscosity conductive inks, extrusion of insulating silicone pastes, and in situ surface activation via cold-air plasma. Image courtesy of Saint-Petersburg State University/University of Sheffield.
Toward Clinical Applications
The researchers have shown that 3D printers can produce implants that can communicate with brains and nerves. Following this early work, they aim to demonstrate how the devices are robust when implanted for long periods. The researchers’ ambition, however, is to translate their work to the clinic and open up the possibilities of personalized medicine to neurosurgeons. Minev and Musienko expressed their desire to see the innovative neural implant technology in the operating theatre. Suggesting that perhaps in the future it will be possible to produce patient-specific neural implants right in the hospital, while the patient is being prepared for surgery.
This vision of on-demand, patient-specific neural implants represents a paradigm shift in neurosurgical practice. Current neural implant procedures require pre-manufactured devices that may not perfectly match a patient’s anatomy. The ability to print implants customized to individual brain or spinal cord structures could improve surgical outcomes and reduce complications.
The potential applications extend beyond paralysis treatment. Researchers foresee applications in:
- Epilepsy monitoring: Custom electrode arrays for precise seizure focus localization
- Parkinson’s disease: Deep brain stimulation electrodes optimized for individual anatomy
- Chronic pain management: Targeted neuromodulation devices
- Stroke rehabilitation: Brain-computer interfaces for motor recovery
- Alzheimer’s disease: Memory circuit stimulation devices
Frequently Asked Questions (FAQ)
What is NeuroPrint technology?
NeuroPrint is a hybrid 3D printing technology developed at Saint-Petersburg State University that enables the rapid production (within 24 hours) of patient-specific neural and muscular implants. It combines ink-jet dispensing of conductive inks, extrusion of silicone insulators, and cold-air plasma activation to create soft, biocompatible neural interfaces that can be customized for individual patients.
How does NeuroPrint differ from traditional neural implant manufacturing?
Traditional neural implant manufacturing typically takes 2-8 weeks, costs significantly more ($5,000-15,000 per prototype), and offers limited customization options. See also: Best Budget 3D Printer Upgrades That Actually Impr…. NeuroPrint reduces production time to approximately 24 hours, costs around $500-1000 per prototype, allows fully patient-specific designs that can be easily modified, and uses soft, biocompatible materials that integrate better with neural tissue than rigid traditional metals.
What medical conditions could NeuroPrint implants treat?
NeuroPrint implants have potential applications for treating multiple neurological conditions including spinal cord injuries, epilepsy, Alzheimer’s disease, Parkinson’s disease, multiple sclerosis, chronic pain, and stroke rehabilitation. The technology enables brain-computer interfaces for paralyzed patients and could provide targeted neuromodulation for various neurological disorders.
Has NeuroPrint been tested on humans?
As of the 2020 publication in Nature Biomedical Engineering, NeuroPrint technology has been tested on animal models including cats, rats, and zebrafish. The research demonstrated long-term integration and functional stability of the printed bioelectronic interfaces. Clinical trials for human use have not yet begun, but the researchers have expressed plans to translate the technology to clinical applications and hospital settings.
What materials are used in NeuroPrint implants?
NeuroPrint implants are constructed using soft, biocompatible materials including silicone for structural support and insulation, platinum microparticles for electrical conductivity, and other conductive elements. The combination of soft composites allows the implants to match the mechanical properties of neural tissue, reducing inflammation and scarring while maintaining electrical functionality for neural stimulation and recording.
How does NeuroPrint compare to Neuralink?
While both technologies aim to create brain-computer interfaces, they differ in approach. Neuralink focuses on high-bandwidth communication using a robotic insertion system for rigid electrode arrays. NeuroPrint emphasizes patient-specific soft implants that can be rapidly prototyped and customized for individual anatomy. Neuralink has begun human trials, while NeuroPrint is in the animal testing phase with plans for clinical translation. The technologies could potentially be complementary rather than competing.
Can NeuroPrint implants be modified after initial production?
Yes, one of the key advantages of NeuroPrint technology is the ability to quickly modify and reprint implant designs. Neuroscientists can test a prototype, request changes, and receive an updated version within 24 hours. This rapid iteration cycle significantly accelerates research and development compared to traditional manufacturing methods where design changes require weeks of re-tooling.
What are the future goals for NeuroPrint technology?
The researchers aim to demonstrate long-term stability of implanted devices and translate the technology to clinical settings. Their vision includes hospital-based 3D printing systems that can produce patient-specific neural implants while a patient is being prepared for surgery. They also plan to expand applications to various neurological conditions and explore the potential for personalized medicine in neurosurgery.
The post NeuroPrint: Scientists Bioprint Brain Implants for Custom Neural Interfaces appeared first on 3DPrint.com | The Voice of 3D Printing / Additive Manufacturing.
from 3DPrint.com | The Voice of 3D Printing / Additive Manufacturing https://bit.ly/3im10KT
Related: Wake Forest Researchers Claim to Bioprint Skeletal Muscle Constructs With Neural · Characterization of Conventionally Manufactured & 3D Printed Acetabular Cup Impl · Researchers Develop Workflow for Repeatable Fabrication of PMMA Craniofacial Imp
📌 Related Articles
- Best 3D Printer Upgrades That Actually Improve Print Quality: Complete 2026 Guide
- Best Budget 3D Printer Upgrades That Actually Improve Print Quality: Belts, Springs, Hotends & More
- Prusa Research Mini+ vs Prusa MK4: Full Specs Comparison & Buyer’s Guide
- ABS 3D Printing Settings Guide: Temperature, Enclosure, and Cooling for Strong Parts
- Bambu Lab P1S vs Bambu Lab P2S: Full Specs Comparison & Buyer’s Guide