Researchers from France and Lebanon performed a unique study into materials science for 3D printing recently, publishing the details in ‘Coumarins as Powerful Photosensitizers for the Cationic Polymerization of Epoxy-Silicones under Near-UV and Visible Light and Applications for 3D Printing Technology.’
Eight chemical compounds known as coumarins (numbered one through eight) were considered for use in the cationic polymerization (CP) of epoxysilicones when combined with 4-isopropyl-4’-methyldiphenyliodonium tetrakis(pentafluorophenyl)borate (IOD). The authors explain that as light-induced cationic polymerization processes become more popular for industrial use, mechanical and chemical resistance properties are expanded, along with better opportunity for ‘low shrinkage.’
In this study, the researchers noted superior reactivity in the new coumarin systems, displaying ‘very high efficiencies’ of the compounds with lower power irradiation sources. Comparisons in the study were made with other common photosensitizers like CPTX and an anthracene derivative known as DBA, along with other commercial coumarins. Ultimately, the authors hoped to use the coumarin-based formulations to 3D print ‘thick 3D polymer samples.’
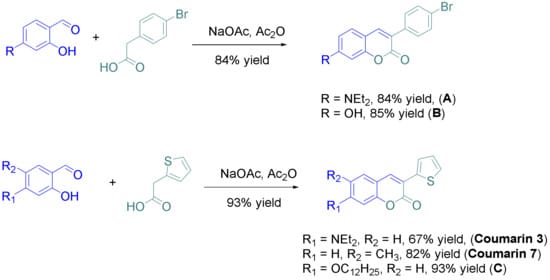
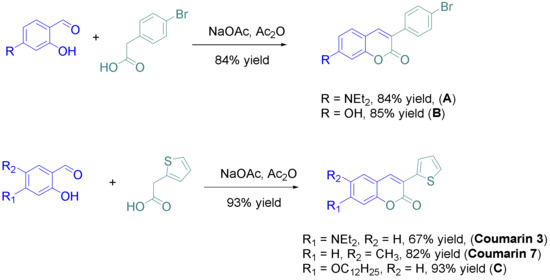
“ … only one procedure was used for the synthesis of the eight derivatives, consisting in the condensation of the appropriate salicylaldehyde derivative with the suitable phenylacetic acid derivative in acetic anhydride. All coumarins A–C and coumarins 3 and 7 could be obtained in yields ranging from 67 to 85%.
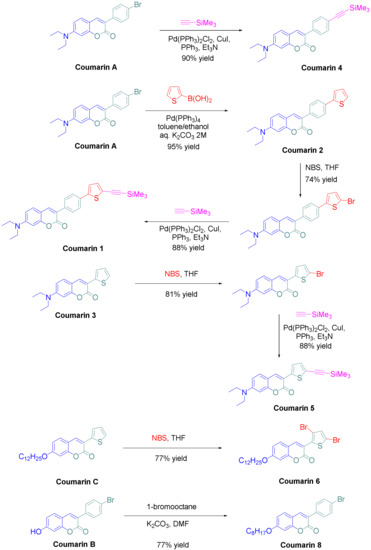
Coumarins numbered 1 and 2, 4-6, and 8 were prepared via synthetic routes.
“Thus, starting from coumarin A, coumarins 2 and 4 could be respectively obtained by a Suzuki cross-coupling reaction with 2-thiopheneboronic acid and a Sonogashira reaction with trimethylsilyl acetylene, providing the two molecules in yields of 95 and 90%, respectively. Coumarin 1 could be obtained in two steps starting from coumarin 2, by bromination of the latter with N-bromo-succinimide (NBS) followed by a Sonogashira reaction with trimethylsilylacetylene,” explained the researchers.
“Coumarin 1 was obtained in pure form in 65% yield for the two steps. Using the same strategy than that used for coumarin 1, coumarin 5 could be prepared in a higher reaction yield, reaching 71% for the two steps. Finally, coumarins 6 and 7 were synthesized in one step, by bromination of coumarin C with two equivalents of N-bromosuccinimide and by alkylation of coumarin B with 1-bromooctane under basic conditions. In turn, coumarins 6 and 7 were isolated in pure form in 77 and 77% yields, respectively. Among the eight coumarins reported in this work, four of them have never been reported in the literature and were specifically designed to act as photoinitiators.”
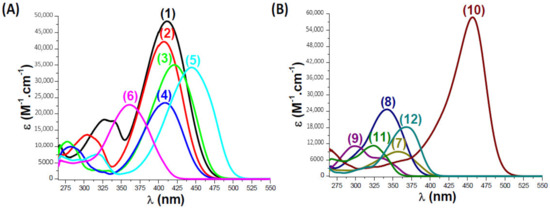
UV-Visible Absorption Spectra of Coumarins Studied in Acetonitrile: (A): (1) Coumarin 1; (2) Coumarin 2; (3) Coumarin 3; (4) Coumarin 4; (5) Coumarin 5; (6) Coumarin 6. (B): (7) Coumarin 7; (8) Coumarin 8; (9) Com. Coum 1; (10) Com. Coum 2; (11) Com. Coum 3; (12) Com. Coum 4, respectively.
Noting that currently, photoinitiators are quite rare for 3D applications, the benefits of using the cationic process include the capability for reducing shrinkage. EPOX-Si200 was used in the experiments, as varying thick 3D patterns were generated by a laser diode. In less than one minute, each 3D pattern was produced under irradiation. The researchers also realized that spatial control was highlighted as polymerization occurred in the irradiated area.
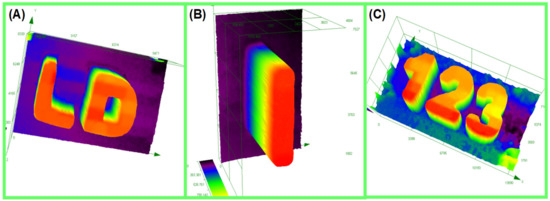
3D Printing Experiments concerning the CP of Epoxysilicones upon irradiation with a Laser Diode (@405 nm): Characterization of the thick 3D generated patterns by numerical optical microscopy: (A) Coumarin 2/IOD (0.04%/0.8% w/w) (thickness = 1800 μm); (B) Coumarin 3/IOD (0.018%/0.36% w/w) (thickness = 1320 μm) and (C) Coumarin 7/IOD (0.05%/1% w/w) (thickness = 2200 μm); respectively.
“These can be interesting alternatives to well-known commercial coumarins or CPTX or DBA/IOD reference systems. The development of new cationic systems for 3D printing applications (upon laser diode @405 nm) was also accomplished in the presence of these coumarin-based PISs. Future development of new high-performance photoinitiating systems are under progress and will be presented in our upcoming papers,” concluded the researchers.
While this study remains quite unique, many researchers have performed studies regarding other ways to incorporate materials and compounds, with molecular 3D printers and other techniques. What do you think of this news? Let us know your thoughts! Join the discussion of this and other 3D printing topics at 3DPrintBoard.com.
[Source / Images: ‘Coumarins as Powerful Photosensitizers for the Cationic Polymerization of Epoxy-Silicones under Near-UV and Visible Light and Applications for 3D Printing Technology’]
The post Cationic Polymerization of Epoxy-Silicones with Coumarin Systems for 3D Printing appeared first on 3DPrint.com | The Voice of 3D Printing / Additive Manufacturing.
from Your daily news from 3DPrint.com https://bit.ly/2Uuaaw7