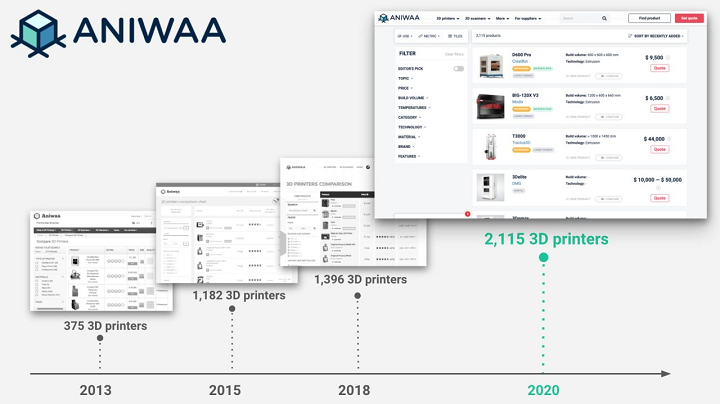
Launched in the summer of 2013 by two childhood friends looking to process and organize all the data of the 3D printing ecosystem, independent online platform Aniwaa simplifies the process of researching, evaluating, and purchasing 3D printers and scanners. French entrepreneurs and Aniwaa co-founders Martin Lansard, CEO, and Pierre-Antoine Arrighi, Technical Advisor, have registered the company in Singapore, with the main team operating remotely out of both Cambodia and France.
There’s a lot of information about industrial 3D printers flying around on the Internet, and not nearly enough user-friendly and reliable tools to help professionals looking to invest in the technology. On top of that, the AM industry is changing constantly, which makes it even more difficult to research and compare the products. Aniwaa is focused on making this comparison process easier for 3D printer users, which is why it’s been working hard to build an even more comprehensive 3D printing system database.
Now, the company has officially launched its new and improved 3D printer comparison platform.
“3D printers are a fast-growing category; we’re seeing many professional applications reaching maturity and a strong untapped potential overall. New hardware is being released frequently, which is creating a very fragmented market space and making it difficult for potential buyers to make an informed choice. That’s why we’re constantly improving our tools to help our users find the best AM hardware for their needs and budget,” Aniwaa co-founder Arrighi stated in a press release.
The Role of 3D Printing in Medicine
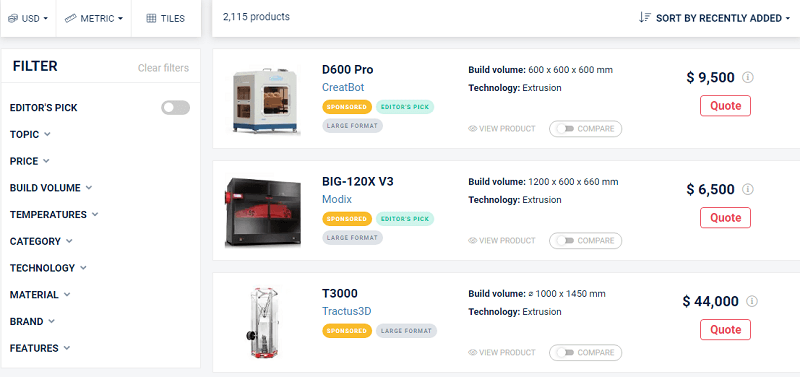
Aniwaa has completely revamped its 3D printer comparison engine, and the redesigned tool indexes the entire spectrum of systems—over 2,100 3D printing products, with more coming all the time. The launch of its updated platform also marks the company’s growing leadership in the AM industry’s product evaluation sector.
“I’m really proud of what we’ve achieved. With the COVID situation, physical trade shows are declining sharply while the product discovery and comparison phases are moving online,” Aniwaa’s other co-founder, Lansard, explained. “This new release strengthens Aniwaa’s position as the go-to additive manufacturing platform, helping not only buyers but also suppliers– from manufacturers aiming to build their brands to resellers looking for qualified leads. We’re all excited by what’s ahead and look forward to supporting the growth of our industry and helping 3D printing companies thrive.”
Biocompatible Materials and Processes
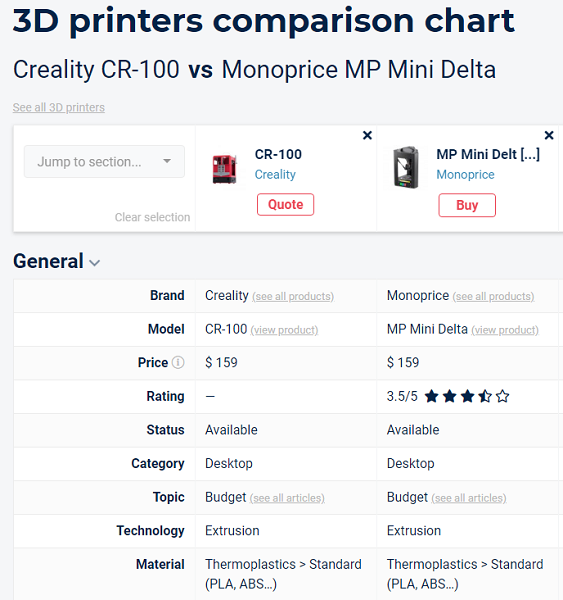
The tool offers multiple comparison, sorting, and filtering options right off the bat. To test out how easy Aniwaa’s new comparison platform was, I tried it out for myself, sorting from low to high prices (I’m not made of money) and ended up selecting two desktop extrusion-based 3D printers—the Creality CR-100 and the Monoprice MP Mini Delta—to compare side by side, though I could have chosen a total of four to benchmark if I’d wished.
In addition to price, rating, status, category, topic, technology, and material, the Aniwaa overview also compares points like printer build size and volume, minimum layer thickness, maximum print speed, nozzle and filament diameter, what kind of connectivity it offers (WiFi, USB, Bluetooth, etc.), and possible features such as dual extruders, a heated print bed, or a pellet extruder.
“Our new tools allow users to easily cut through the clutter and identify the right 3D printers or 3D scanners among the thousands available on the market,” Lansard stated in a LinkedIn post about the launch.
Clinical Applications and Case Studies
“The product evaluation phase of the purchase funnel is quickly moving to online across all sectors, and the AM and 3D capture industries are no exceptions. See also: Best Budget 3D Printer Upgrades That Actually Impr…. We bet on that trend early and today we’re proud that Aniwaa has established itself as the go-to platform where professional buyers research and compare products before making a purchase decision.”
This new release also signifies Aniwaa’s move to a full platform model. In addition to updated 3D printer comparison, the company also offers marketing and selling solutions in order to help different brands connect with professional buyers, and connects these possible buyers with resellers from its global network.
(Source/Images: Aniwaa)
Regulatory Considerations and Safety
The post Aniwaa Launches New and Improved 3D Printer Comparison Platform appeared first on 3DPrint.com | The Voice of 3D Printing / Additive Manufacturing.
from Your daily news from 3DPrint.com https://bit.ly/2Fv0xZ7
Related Articles
Frequently Asked Questions
How is 3D printing used in medicine?
3D printing is used in medicine for surgical planning models, custom implants, bioprinting tissue scaffolds, drug delivery systems, dental aligners, and prosthetics. It enables patient-specific solutions that improve outcomes and reduce surgery time.
What materials are biocompatible for 3D printing?
Common biocompatible materials include PEEK, titanium alloys (Ti6Al4V), bio-ceramics (hydroxyapatite), medical-grade resins, PLA for temporary implants, and hydrogels for bioprinting. Material choice depends on the application and required mechanical properties.
Is 3D printed medical equipment FDA approved?
Yes, several 3D printed medical devices have FDA clearance, including orthopedic implants, dental restorations, and surgical guides. Each device must go through the appropriate regulatory pathway based on its risk classification.
📌 Related Articles
- Best 3D Printer Upgrades That Actually Improve Print Quality: Complete 2026 Guide
- Best Budget 3D Printer Upgrades That Actually Improve Print Quality: Belts, Springs, Hotends & More
- Creality Creality Ender 3 V3 Plus vs Creality Ender 3 Pro: Full Specs Comparison & Buyer’s Guide
- Prusa Research Mini+ vs Prusa MK4: Full Specs Comparison & Buyer’s Guide
- ABS 3D Printing Settings Guide: Temperature, Enclosure, and Cooling for Strong Parts