Frequently Asked Questions
What is china: 3d printed drug delivery scaffolds, evaluated in rat femoral bones?
Quick Answer: What are 3D Printed Drug Delivery Scaffolds for Bone Regeneration. This topic is increasingly relevant in the 3D printing community as the technology continues to advance and become more accessible to hobbyists and professionals alike.
Disclosure: As an Amazon Associate, 3dput.com earns from qualifying purchases. This supports our independent testing and reviews.
How does this relate to 3D printing?
This topic is directly connected to additive manufacturing and 3D printing technology. Understanding these concepts helps improve print quality, expand capabilities, and explore new applications for desktop and industrial 3D printing.
What are the practical applications?
The practical applications span from rapid prototyping and custom manufacturing to educational projects and functional parts production. These techniques can be applied with most consumer-grade and professional 3D printers available today.
Quick Answer: What are 3D Printed Drug Delivery Scaffolds for Bone Regeneration?
3D printed drug delivery scaffolds are advanced biomaterials designed to support bone regeneration while releasing therapeutic compounds in a controlled manner. Chinese researchers have developed scaffolds made from 1393 beta-tricalcium phosphate coated with mesoporous bioactive glass (MBG) that can load and release drugs like dexamethasone (DEX), bone morphogenetic protein-2 (BMP-2), and DNA. These scaffolds provide structural support for bone tissue growth while delivering bioactive agents that enhance osteogenic differentiation and bone formation. The study demonstrated that MBG-coated scaffolds loaded with BMP-2 significantly improved bone regeneration in rat femoral defects over 12 weeks, showing promise for treating bone injuries and diseases in clinical applications [1].
Chinese researchers are working to improve drug delivery systems for bone repair, detailing their findings in recently released ‘A novel vehicle-like drug delivery 3D printing scaffold and its applications for a rat femoral bone repairing in vitro and in vivo’ [1].
Bone regeneration continues to be an area of challenge for researchers and doctors as they attempt to treat patients and improve their quality of life overall, performing a wide range of studies regarding materials, properties, and methods. Biomaterials are in great demand, and authors point out that this demand increases as issues like traffic injuries rise, along with natural disasters and other catastrophes delivering patients in need to hospitals [2].
The Challenge of Bone Regeneration
Bone regeneration remains one of the most challenging areas in orthopedic medicine and tissue engineering. Each year, millions of people worldwide suffer from bone fractures, defects, or degenerative bone diseases that require surgical intervention and bone grafting. Traditional treatments, such as autografts (using patient’s own bone) or allografts (donor bone), have limitations including donor site morbidity, limited availability, and risk of disease transmission [2].
The development of synthetic scaffolds represents a critical advancement in bone tissue engineering. These scaffolds serve as temporary extracellular matrices that guide cell attachment, proliferation, and differentiation while gradually degrading as new bone tissue forms. For bone repair to be successful, scaffolds must possess specific properties:
- Pore architecture: Optimized pore size and interconnectivity to allow cell infiltration, vascularization, and nutrient diffusion
- Mechanical strength: Sufficient compressive strength to support bone defects during healing while matching the mechanical properties of native bone
- Bioactivity: Ability to chemically bond with bone tissue through surface reactions that promote mineralization
- Degradation: Controlled degradation rate that matches new bone formation, avoiding premature scaffold collapse or long-term foreign body presence
- Controllable drug-delivery ability: Capacity to load and release therapeutic agents with controlled kinetics for enhanced regeneration
Mesoporous Bioactive Glass (MBG) Technology
Mesoporous bioactive glass (MBG) has emerged as one of the most promising bone regenerative materials due to its well-acknowledged bioactivity, biodegradability, and osteoconductivity. Unlike traditional bioactive glass, MBG features a mesoporous structure with pore sizes ranging from 2-50 nm, providing an exceptionally high surface area for drug loading and protein adsorption [3].
According to recent comprehensive reviews, mesoporous bioactive glasses have significantly expanded their functional applications beyond those of traditional bioactive glasses. Currently, MBGs are utilized in regenerative medicine, bone regeneration as scaffolds and coatings, tissue engineering as 3D supports, wound healing, gene delivery, controlled drug delivery systems, antimicrobial applications, and bioimaging [3].
3D printed MBG scaffolds obtained through advanced fabrication techniques possess remarkable properties:
- High mechanical strength: Approximately 200 times greater than traditional polyurethane foam-templated MBG scaffolds
- Controllable pore architecture: Precise control over pore size, shape, and interconnectivity through 3D printing parameters
- Excellent hydroxylapatite (HA) mineralization ability: Rapid formation of bone-like apatite layers when exposed to physiological fluids
- Potential for bone tissue grafting: Bioactive glass releases silicon and calcium ions that stimulate bone formation
Research published in Nature Scientific Reports demonstrated that functionalized mesoporous bioactive glass scaffolds (N-MBGS) have an interconnected macroporous network that is helpful for cell adhesion and migration, while the mesopores enhance bioactivity and further promote cell adhesion [4]. The study confirmed that the mesopore structures in MBG scaffolds play an important role in improving loading efficiency, decreasing burst effects, and maintaining the bioactivity of therapeutic agents [5].
However, while bioglass offers excellent bioactivity and releases beneficial ions for bone regeneration, it still lacks the necessary versatility required for complex drug delivery systems. The challenge lies in achieving controlled, sustained release of multiple therapeutic agents with different molecular weights and release kinetics [6].
1393 Beta-Tricalcium Phosphate Scaffolds
The foundation of this drug delivery system is 1393 beta-tricalcium phosphate (β-TCP), a well-established biomaterial in bone tissue engineering. Recent reviews in Frontiers in Materials have highlighted that β-TCP has emerged as a promising material for bone regeneration applications due to its osteoconductivity and osteoinductivity properties as well as its superior degradation in vivo [2]. 1393 is known for its:
- Biocompatibility: Excellent compatibility with bone tissue and cells
- Biodegradability: Gradual resorption as new bone forms
- Osteoconductivity: Ability to support bone cell attachment and growth
- Chemical stability: Maintains structural integrity in physiological environments
Porous 3D printed 1393 scaffolds have attracted significant research attention. Studies published in Nature Scientific Reports have demonstrated that β-TCP scaffolds are widely employed in bone tissue regeneration applications [7]. Materials like hydroxylapatite (HA) and beta-tricalcium phosphate (β-TCP) offer potential to chemically bond with host bone through bioactive interactions [2].
Research has shown that β-TCP granules can improve osteogenesis in vitro and establish innovative osteo-regenerators for bone tissue engineering in vivo [8]. These scaffolds have been successfully used as BMP-2 delivery systems for bone tissue engineering, with studies confirming the activity of BMP-2 released from the scaffolds and bone formation within scaffold pores [9].
Innovative MBG Coating Strategy
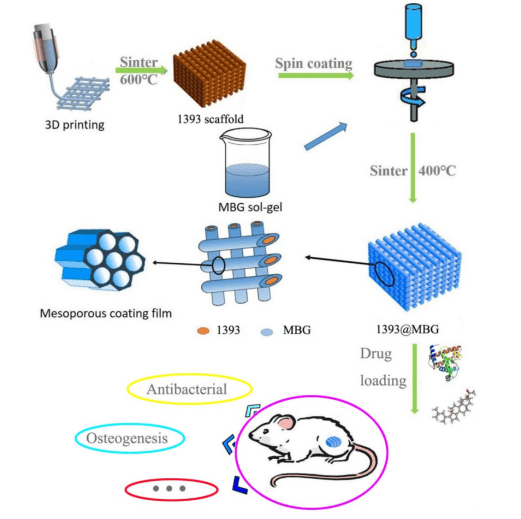
The key innovation in this research is the application of MBG coating onto the surface of 1393 scaffolds. This approach creates a novel vehicle-like drug delivery system that combines the mechanical support of 1393 with the bioactivity and drug-loading capacity of MBG [1].
“Herein, applying a mesoporous bioactive glass [MBG] coating onto the surface is a viable option for drug delivering capacity of bioactive glass scaffolds,” stated researchers. “With mesopores on the surface, DNA, cytokines and some drugs can be loaded onto the scaffold, meeting the requirements of disease treatment or further enhanced bone regeneration.” [1]
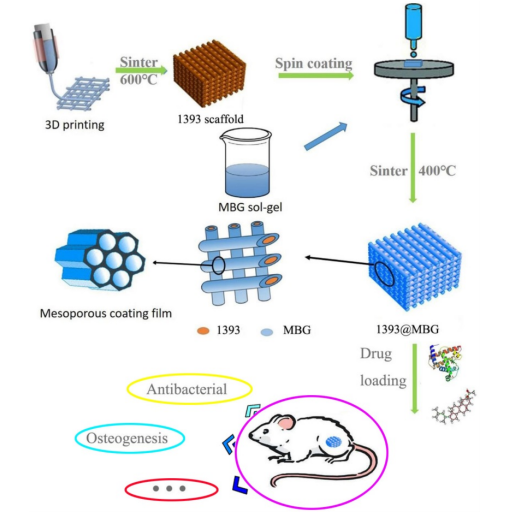
The flow chart of fabricating a novel drug delivery 1393@MBG scaffold built by 3D printing technology for bone repairing.
Mechanism of Drug Loading
The mesoporous structure of MBG enables efficient drug loading through various mechanisms:
- Adsorption: Large surface area allows physical adsorption of drugs and proteins onto the MBG surface
- Capillary action: Mesopores (2-50 nm) draw liquid solutions containing therapeutic agents into the coating
- Electrostatic interactions: Surface charge properties of MBG facilitate binding of charged molecules
This multi-modal loading capacity allows researchers to incorporate diverse therapeutic agents with different chemical properties and molecular sizes into a single scaffold system. The mesopore structures in MBG scaffolds play an important role in improving loading efficiency, decreasing the burst effect and maintaining the bioactivity of therapeutic agents [5].
Drug Selection: DEX, BMP-2, and DNA
The research focused on three different therapeutic agents, each serving distinct purposes in bone regeneration:
Dexamethasone (DEX)
Dexamethasone is a synthetic glucocorticoid with potent anti-inflammatory and immunosuppressive properties. In bone tissue engineering, DEX is used to:
- Reduce inflammation at implantation site
- Suppress immune rejection reactions
- Modulate cellular behavior for enhanced regeneration
The drug’s chemical structure and molecular weight allow for controlled release from the MBG coating, making it suitable for managing the acute inflammatory phase of bone healing.
Bone Morphogenetic Protein-2 (BMP-2)
BMP-2 is a growth factor that plays a crucial role in bone formation and repair. According to comprehensive reviews in PMC, bone morphogenetic proteins (BMPs) are responsible for bone formation during embryogenesis and bone regeneration and remodeling [10]. As one of the most potent osteoinductive factors, BMP-2:
- Is commercially approved by FDA for bone tissue regeneration applications
- Promotes osteoblastic differentiation of mesenchymal stem cells into bone-forming cells
- Enhances bone formation in the early phase of regeneration
- Upregulates osteogenesis-related genes including alkaline phosphatase (ALP), osteopontin (OPN), and runt-related transcription factor 2 (Runx2) [11]
Studies have shown that BMP-2 can achieve effective bone regeneration with remarkably low doses—as low as 1 μg in rat femoral defects, compared to previously reported minimum thresholds of 11 μg [1]. Research published in PMC demonstrated that BMP-2 enhanced bone repair after injury, as well as stimulating an increase in osteoblast differentiation and bone regeneration [11]. This makes BMP-2 particularly valuable for controlled release systems where maximizing therapeutic effect while minimizing side effects is crucial.
Recent clinical reviews have examined the current status of the clinical application and efficacy of recombinant human BMP-2 (rhBMP-2) in maxillofacial surgery, noting that its osteoinductive ability was verified in animal models before evaluation in human clinical trials [12]. The aims of these studies include examining its safety and limitations through literature reviews [12].
DNA Loading
The inclusion of DNA in the drug delivery system serves multiple purposes:
- Genetic material delivery: Potential for gene therapy applications in bone regeneration
- Cytokine loading: DNA can serve as a model or carrier for various cytokines and signaling molecules
- Release kinetics comparison: Provides another data point for understanding MBG’s multi-drug loading capacity
DNA loading demonstrated the versatility of the MBG coating to accommodate molecules with different sizes and charges, expanding the potential applications of the scaffold system [3].
Scaffold Fabrication and Characterization
The research employed advanced 3D printing techniques to fabricate the 1393 scaffolds with precise control over architecture. The fabrication process included:
- 1393 powder preparation: Using 1393 β-TCP powder with controlled particle size distribution
- MBG coating application: Applying mesoporous bioactive glass layer onto scaffold surfaces
- 3D printing: Using layer-by-layer deposition to create porous structures with designed geometries
- Drug loading: Immersing scaffolds in solutions containing DEX, BMP-2, or DNA

FESEM images of (a, c) as fabricated 1393 and 1393@MBG scaffold; (b, d) the cross section of as fabricated 1393 and 1393@MBG scaffold.
Characterization Techniques
The fabricated scaffolds underwent comprehensive characterization using various analytical techniques:
- Field Emission Scanning Electron Microscopy (FESEM): To examine surface morphology and pore structure at high resolution
- Nitrogen adsorption-desorption isotherms: To determine specific surface area and pore size distribution of MBG powders [5]
- Simulated Body Fluid (SBF) immersion: To evaluate bioactivity and mineralization potential
- Ninhydrin staining: To assess drug release profiles using colorimetric detection

(a, b) FESEM images and surface profile of as fabricated 1393 scaffold; (c, d) FESEM image and surface profile of as fabricated 1393@MBG scaffold; (e, f) FESEM image and surface profile of as fabricated 1393@MBG scaffold after immersed scaffold; (g, h) FESEM image and surface profile of as fabricated 1393@MBG scaffold after immersed scaffold; (i) The Ra of 1393 and 1393@MBG scaffold surface when immersed from 0 to 90 days; (j) The compressive strength of 1393 and 1393@MBG scaffold on day 0, 30 and 90. mean ± SD, n = 5. *Significant difference when compared to 1393 (p < 0.05).
Drug Release Assessment
After immersion in simulated body fluids (SBF), drug release was assessed using ninhydrin staining, with detection values at:
- 241 nm for DEX: Dexamethasone release measured at specific wavelength
- 260 nm for DNA: DNA release measured at different wavelength
- 567 nm for BMP-2: Bone morphogenetic protein-2 release detected at its characteristic wavelength
This multi-wavelength detection approach allowed researchers to simultaneously monitor the release kinetics of all three therapeutic agents from the scaffold system, demonstrating MBG’s capacity for multi-drug delivery. The mesopore structures in MBG scaffolds play an important role in maintaining the bioactivity of therapeutic agents [5].
In Vitro and In Vivo Evaluation
The comprehensive study evaluated the scaffold system both in controlled laboratory conditions (in vitro) and in a living organism (in vivo) model using human bone marrow mesenchymal stem cells (hBMSCs) and rat femoral bone defects [1].
In Vitro Studies with hBMSCs
Human bone marrow mesenchymal stem cells were cultured with the scaffolds to assess:
- Cell proliferation: Ability of scaffolds to support stem cell growth and expansion
- Alkaline Phosphatase (ALP) activity: Marker of early osteogenic differentiation
- Osteogenic gene expression: Upregulation of bone formation-related genes
The results showed that MBG-coated scaffolds enhanced both the proliferation and ALP activity of hBMSCs, while upregulating osteogenesis-related genes. Research on functionalized MBG scaffolds confirmed that the interconnected macroporous network is helpful for cell adhesion and migration, and that mesopores enhance bioactivity and further promote cell adhesion [4]. This indicates that the MBG coating improves the biological performance of 1393 scaffolds, making them more conducive to bone cell differentiation and activity.
In Vivo Rat Femoral Defect Model
Critical-size femoral defects were created in rats to evaluate bone regeneration in a living system. The 1393@MBG scaffolds loaded with BMP-2 were implanted and monitored over 12 weeks post-implantation.
- Defect size: Critical-size defects that do not heal spontaneously in rats
- Implantation period: 12 weeks for comprehensive bone regeneration assessment
- Control groups: Comparison with untreated defects and scaffolds without drug loading
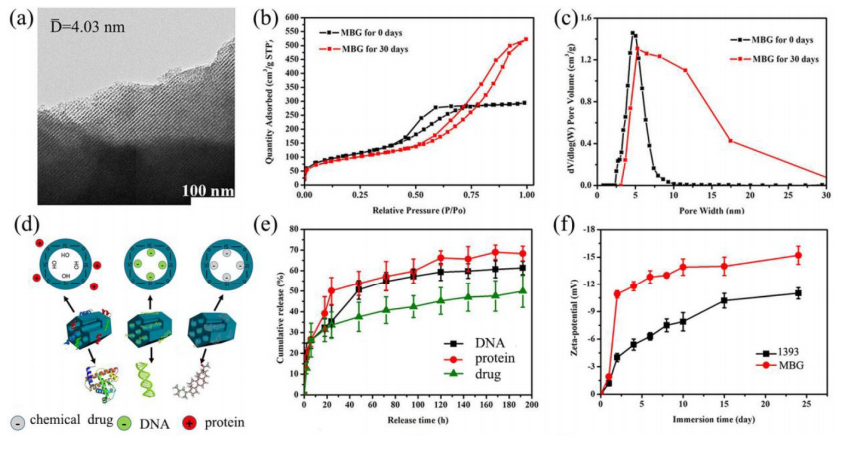
(a) TEM image of as made MBG powder; (b) N2 adsorption–desorption isotherms and (c) corresponding pore size distributions of MBG powders; (d) illustrator of mechanism of MBG loading protein, DNA and chemical drug; (e) chemical drug, DNA and protein release profiles from MBG powders in SBF at 37 ℃; (f) Zeta-potential of 1393 and MBG glass powder immersed in SBF at 37 °C as a function of immersion time; mean ± SD, n = 5.
Results and Outcomes
The study reflected positive results, with MBG proving its capacity to both load and control drug release. BMP-2 exhibited the most rapid rate of release among the three agents, followed by DNA, and then DEX. This differential release kinetics allows researchers to design multi-phase therapeutic regimens where different agents act at different stages of healing [1].
“The main group of MBG was silicon oxide group, thus when immersed in a water solution the surface of MBG will exhibit a positive charge which will more easily absorb negative materials such as –OH groups, DEX and DNA,” stated authors. “So, this may explain the phenomenon that BMP-2 release was better than that of DNA and DEX, in order, BMP-2 > DNA > DEX.” [1]
The prepared BMP-2 loaded 1393@MBG scaffolds significantly improved bone regeneration in osseous defects at 12 weeks post-implantation, demonstrating the therapeutic potential of the system [1]. Notably, the low BMP-2 dose (1 μg) achieved effective healing, highlighting the efficiency of the controlled release system. Studies on BMP-2 delivery using β-TCP scaffolds have confirmed similar results, with histological observations confirming bone formation within BMP-2-loaded scaffold pores [9].
Comparison: Scaffold Materials for Bone Regeneration
| Material | Key Properties | Advantages | Limitations | Best Applications |
|---|---|---|---|---|
| Mesoporous Bioactive Glass (MBG) | High bioactivity, mesoporous (2-50 nm), high surface area [3] | Excellent drug loading, ion release, HA mineralization [5] | Limited mechanical strength alone | Drug delivery, coating applications, enhanced regeneration [3] |
| 1393 Beta-Tricalcium Phosphate (β-TCP) | Biodegradable, osteoconductive, chemically stable [2] | Good mechanical support, proven biocompatibility | Slow degradation, limited bioactivity alone | Structural scaffolding, bone defects, dental applications [7] |
| Hydroxylapatite (HA) | Similar to bone mineral, excellent biocompatibility | Direct bonding with host bone | Brittle, slow degradation | Filler material, dental implants, coatings |
| Bioactive Glass | Bioactive, releases Si and Ca ions | Promotes bone formation, proven clinical use | Lacks drug delivery versatility [6] | Bone grafting, dental implants, coatings |
| Polymer Scaffolds (PCL, PLA) | Tunable degradation, easy processing | Flexible, customizable, easy to print | Low mechanical strength, hydrophobic | Temporary support, soft tissue engineering |
Comparison: Drug Delivery Methods for Bone Regeneration
| Method | Release Mechanism | Advantages | Disadvantages | Therapeutic Agents |
|---|---|---|---|---|
| MBG Coating on 1393 Scaffold | Adsorption, desorption, electrostatic interactions [1] | Multi-drug loading, controlled release, enhanced bioactivity [5] | Complex fabrication, coating uniformity challenges | DEX, BMP-2, DNA, cytokines [3] |
| Direct Scaffold Loading | Diffusion from polymer matrix | Simple fabrication, good for large molecules | Burst release, limited multi-drug capability | Growth factors, antibiotics |
| Injectable Hydrogels | Swelling/degradation controlled release | Minimally invasive, fills irregular defects | Limited mechanical strength, rapid clearance | BMP-2, anti-inflammatories |
| Nanoparticle Encapsulation | Surface degradation, diffusion | Precise control, multi-drug possible | Complex manufacturing, potential toxicity | Various proteins, drugs |
| Microsphere Embedding | Polymer degradation, diffusion | Established technology, extended release | Burst release, limited loading capacity | Antibiotics, growth factors |
Future Directions and Clinical Applications
The success of this drug delivery scaffold system suggests promising avenues for clinical translation. The combination of structural support, controlled drug release, and enhanced bioactivity addresses multiple challenges in bone regeneration:
Potential Clinical Applications
- Traumatic bone injuries: Acute fractures from accidents or sports injuries
- Spinal fusion surgery: Promoting bone growth between vertebrae [12]
- Dental bone defects: Alveolar ridge augmentation and sinus lifts
- Large segmental bone defects: Resections due to tumors or infection
- Osteoporotic fracture repair: Enhancing healing in compromised bone
Advantages Over Traditional Approaches
The 1393@MBG scaffold system offers several advantages compared to traditional bone grafting and other synthetic options:
- Reduced BMP-2 dose: Controlled release allows effective healing with 1 μg vs. 11 μg threshold [1]
- Multi-drug capability: Single scaffold can deliver anti-inflammatory (DEX), osteogenic (BMP-2), and genetic (DNA) agents [3]
- Enhanced bioactivity: MBG coating promotes HA mineralization and ion release [5]
- Customizable architecture: 3D printing enables patient-specific scaffold geometries
Best 3D Printing Products for Bone Tissue Engineering
For researchers and professionals working in bone tissue engineering, here are recommended products available on Amazon:
3D Bioprinters – Advanced printers for cell-laden bioink fabrication in tissue engineering.
Bone Tissue Engineering Materials – Biomaterials, hydrogels, and scaffolds for bone regeneration research.
Hydroxylapatite 3D Printing Powder – HA powders and filaments for bone tissue engineering applications.
Beta-Tricalcium Phosphate Powder – β-TCP materials for bone regeneration scaffolds.
Stem Cell Culture Supplies – Media, growth factors, and incubation equipment for hBMSC research.
Biomaterials Characterization Equipment – SEM, BET analyzers, and material analysis tools.
Frequently Asked Questions (FAQ)
Q: What are 3D printed drug delivery scaffolds?
3D printed drug delivery scaffolds are three-dimensional porous structures designed to support bone tissue regeneration while releasing therapeutic agents in a controlled manner. These scaffolds are fabricated using 3D printing technologies to create precise pore architectures that allow cell infiltration, nutrient diffusion, and vascularization. They are typically made from biocompatible materials like 1393 beta-tricalcium phosphate, hydroxylapatite, or biodegradable polymers [2]. Drug loading can be achieved through direct incorporation into the scaffold material, surface coating with mesoporous bioactive glass, or embedding drug-containing microspheres [3].
Q: What is mesoporous bioactive glass (MBG)?
Mesoporous bioactive glass (MBG) is a type of bioactive glass featuring a mesoporous structure with pore sizes ranging from 2-50 nm [3]. This mesoporosity provides an exceptionally high surface area for drug loading and protein adsorption compared to conventional bioactive glass. MBG exhibits excellent bioactivity, biodegradability, and osteoconductivity, making it ideal for bone regeneration applications [3]. According to recent reviews, MBGs are utilized in regenerative medicine, bone regeneration as scaffolds and coatings, tissue engineering as 3D supports, wound healing, gene delivery, controlled drug delivery systems, and antimicrobial applications [3]. When implanted, MBG releases silicon and calcium ions that stimulate bone formation and promotes hydroxylapatite (HA) mineralization on its surface [5].
Q: What are the advantages of coating 1393 scaffolds with MBG?
Coating 1393 scaffolds with MBG combines the mechanical support and biocompatibility of 1393 with the superior bioactivity and drug-loading capacity of MBG. Research on functionalized MBG scaffolds demonstrated that the interconnected macroporous network is helpful for cell adhesion and migration, and that mesopores enhance bioactivity and further promote cell adhesion [4]. The MBG coating enables multi-drug loading through its mesoporous structure, provides controlled release kinetics, and enhances osteogenic properties [5]. Additionally, the MBG surface exhibits positive charge in aqueous solutions, facilitating adsorption of negatively charged molecules like DNA, proteins, and certain drugs [1]. This approach creates a synergistic system where the 1393 core provides structural integrity while the MBG coating handles therapeutic delivery and bioactivity enhancement.
Q: What drugs were used in this study and why?
The study used three different therapeutic agents: dexamethasone (DEX), bone morphogenetic protein-2 (BMP-2), and DNA [1]. For more on this topic, see our guide on Best Budget 3D Printer Upgrades That Act…. DEX is an anti-inflammatory drug used to manage inflammation at the implantation site and modulate cellular behavior. BMP-2 is a growth factor commercially approved by FDA for bone tissue regeneration; it promotes osteoblastic differentiation of mesenchymal stem cells and enhances bone formation [11]. DNA was included to demonstrate the versatility of the MBG coating for loading different molecular types and has potential applications in gene therapy [3]. The combination allows for multi-phase therapeutic approaches addressing different aspects of bone healing, with BMP-2 achieving effective bone regeneration at remarkably low doses (1 μg compared to 11 μg threshold) [1].
Q: What are the key properties required for successful bone regeneration scaffolds?
Successful bone regeneration scaffolds must possess several critical properties: optimal pore architecture (pore size 100-500 μm with interconnectivity for cell infiltration and vascularization), sufficient mechanical strength to support the defect site during healing, bioactivity (ability to chemically bond with bone tissue and promote mineralization), controlled biodegradability (degradation rate matching new bone formation), and controllable drug-delivery ability for enhanced regeneration [2]. Additionally, scaffolds should be biocompatible, osteoconductive (supporting bone cell attachment and growth), and have appropriate surface properties for protein adsorption and cell adhesion. Recent reviews highlight that β-TCP has emerged as a promising material for bone regeneration due to its osteoconductivity and osteoinductivity properties as well as its superior degradation in vivo [2].
Q: How does BMP-2 work in bone regeneration?
BMP-2 (bone morphogenetic protein-2) is a potent osteoinductive growth factor that plays a crucial role in bone formation [10]. When delivered to bone defects, BMP-2 binds to receptors on mesenchymal stem cells and initiates signaling pathways that drive osteoblastic differentiation—converting stem cells into bone-forming cells [11]. It upregulates expression of osteogenesis-related genes including alkaline phosphatase (ALP), osteopontin (OPN), and runt-related transcription factor 2 (Runx2) [11]. BMP-2 is commercially approved by FDA for bone tissue regeneration and has shown effectiveness at remarkably low doses (1 μg in rat femoral defects compared to 11 μg threshold for healing in previous models) [1]. Recent clinical reviews have examined the current status of rhBMP-2 in maxillofacial surgery, noting its osteoinductive ability verified in animal models and human clinical trials [12].
Q: What is the difference between in vitro and in vivo studies?
In vitro studies are conducted in controlled laboratory conditions outside a living organism, typically using cell cultures to assess biological responses, toxicity, and basic mechanisms. In vivo studies are performed within a living organism to evaluate actual biological performance, efficacy, and safety in a complex physiological environment. This research employed both approaches: in vitro studies using human bone marrow mesenchymal stem cells (hBMSCs) to evaluate cell proliferation, alkaline phosphatase activity, and gene expression [1]; and in vivo studies using a rat femoral defect model to assess actual bone regeneration over 12 weeks [1]. The combination provides comprehensive understanding of scaffold performance from cellular to organismal levels. Research on β-TCP granules also employed both in vitro evaluation (osteoogenesis) and in vivo assessment (bone tissue engineering) [8].
Q: What are the potential clinical applications of this scaffold system?
The 1393@MBG drug delivery scaffold system has numerous potential clinical applications including traumatic bone injuries (acute fractures from accidents or sports), spinal fusion surgery (promoting bone growth between vertebrae) [12], dental bone defects (alveolar ridge augmentation and sinus lifts), large segmental bone defects (resections due to tumors or infection), and osteoporotic fracture repair (enhancing healing in compromised bone). The system’s advantages—reduced BMP-2 dose requirements (1 μg vs 11 μg threshold) [1], multi-drug delivery capability [3], enhanced bioactivity through MBG coating [5], and customizable 3D printed architecture—make it particularly attractive for clinical translation where precision, efficiency, and patient-specific solutions are valued. Recent clinical reviews have examined rhBMP-2 applications in maxillofacial surgery, confirming its verified osteoinductive ability in clinical trials [12].
Q: How does 3D printing improve scaffold fabrication for bone regeneration?
3D printing offers several advantages for scaffold fabrication including precise control over pore architecture (size, shape, and interconnectivity can be programmed into the digital model and reproduced consistently), customization potential (patient-specific scaffolds can be printed based on medical imaging data), complex geometries (impossible-to-manufacture internal structures can be created), and reproducibility (multiple scaffolds with identical properties can be produced). For bone regeneration applications, 3D printing allows researchers to design scaffolds with optimized pore sizes for cell infiltration and vascularization, create graded structures with varying properties, and incorporate multiple materials or coatings in a single fabrication process. Reviews on mesoporous bioactive glasses highlight that 3D printing enables the fabrication of hierarchical systems with multiscale porosity that enhance osteoconductivity, antibacterial performance, and mineralization capacity [6].
Q: What are the limitations of current bone regeneration approaches?
Current bone regeneration approaches face several limitations: autografts (patient’s own bone) cause donor site morbidity, have limited availability, and variable quality; allografts (donor bone) risk disease transmission and immune rejection; synthetic materials often lack bioactivity without additional coatings, have limited drug delivery capability, and may not match mechanical properties of native bone; BMP-2 therapy requires high doses (11 μg threshold) with potential side effects and high cost [12]; and traditional scaffolds cannot deliver multiple drugs with controlled kinetics or provide enhanced bioactivity [5]. The 1393@MBG coating approach addresses many of these limitations by providing drug loading capacity, controlled release, and enhanced bioactivity while maintaining mechanical support. Recent reviews note that the main group of MBG enables the loading of DNA, cytokines and drugs onto scaffolds, meeting requirements for disease treatment and enhanced bone regeneration [1].
Q: How was drug release evaluated in this study?
Drug release was evaluated using simulated body fluid (SBF) immersion to mimic physiological conditions and ninhydrin staining for colorimetric detection [1]. After scaffolds were immersed in SBF, samples were taken at predetermined time points to measure release of DEX, DNA, and BMP-2. Each drug was detected at a specific wavelength: 241 nm for DEX, 260 nm for DNA, and 567 nm for BMP-2. This multi-wavelength approach allowed simultaneous monitoring of all three agents, revealing that BMP-2 exhibited the most rapid release rate, followed by DNA, then DEX [1]. The surface charge of MBG (positive when immersed) influenced drug adsorption, explaining why negatively charged molecules like BMP-2 showed better loading and release compared to DNA and DEX [1]. Research on MBG drug delivery confirmed that mesopore structures play an important role in improving loading efficiency, decreasing burst effects, and maintaining bioactivity of therapeutic agents [5].
Conclusion
Chinese researchers have developed a promising drug delivery scaffold system that integrates 3D printed 1393 beta-tricalcium phosphate with mesoporous bioactive glass coating for enhanced bone regeneration [1]. The 1393@MBG scaffold successfully loaded and released multiple therapeutic agents—dexamethasone, BMP-2, and DNA—with controlled kinetics, demonstrating the versatility and efficiency of this approach.
In vitro evaluation with human bone marrow mesenchymal stem cells showed that MBG coating enhanced cell proliferation, alkaline phosphatase activity, and osteogenic gene expression [1]. Research on functionalized MBG scaffolds confirmed that the interconnected macroporous network is helpful for cell adhesion and migration, and mesopores enhance bioactivity and further promote cell adhesion [4]. In vivo studies in rat femoral defects demonstrated that BMP-2 loaded scaffolds significantly improved bone regeneration at 12 weeks post-implantation, with remarkably low BMP-2 doses (1 μg) achieving effective healing [1].
This research represents a significant advancement in bone tissue engineering, addressing key challenges in drug delivery, scaffold bioactivity, and controlled release kinetics. Recent comprehensive reviews have highlighted that MBGs have significantly expanded functional applications beyond traditional bioactive glasses, including regenerative medicine, bone regeneration as scaffolds and coatings, tissue engineering, gene delivery, controlled drug delivery systems, and antimicrobial applications [3]. Studies have confirmed that mesopore structures in MBG scaffolds play an important role in improving loading efficiency, decreasing burst effects, and maintaining bioactivity of therapeutic agents [5].
The combination of 3D printing technology, MBG coating strategy, and multi-drug loading capability offers a promising pathway toward clinical applications for treating bone injuries, spinal fusion, dental defects, and other orthopedic conditions requiring enhanced regeneration. As this technology moves toward clinical translation, it holds potential to improve patient outcomes by providing more effective, efficient, and personalized solutions for bone repair and regeneration, reducing the need for high BMP-2 doses and minimizing complications associated with current treatment approaches.

The attachment of hBMSCs on BMP-2 loaded 1393 scaffolds (a) and 1393@MBG scaffolds (b) after culturing for 2 days; The live (green)/dead (red) staining for 1393 (c) and 1393@MBG scaffold (d) immersion solution.
References:
[1] Zhang et al. “A novel vehicle-like drug delivery 3D printing scaffold and its applications for a rat femoral bone repairing in vitro and in vivo.” International Journal of Biological Sciences, 2020. https://www.ijbs.com/v16p1821.pdf
[2] Zhang et al. “Current Application of Beta-Tricalcium Phosphate in Bone Repair and Its Mechanism to Regulate Osteogenesis.” Frontiers in Materials, 2021. https://www.frontiersin.org/journals/materials/articles/10.3389/fmats.2021.698915/full
[3] El-Rashidy et al. “Mesoporous Bioactive Glasses: A Review on Structure-Directing-Based Synthesis, Characterization, and Biomedical Applications.” Materials, 2024. https://www.mdpi.com/1996-1944/19/5/876
[4] Wu et al. “Functionalized mesoporous bioactive glass scaffolds for enhanced bone tissue regeneration.” Scientific Reports, 2016. https://www.nature.com/articles/srep19361
[5] Wu et al. “Mesoporous bioactive glasses: structure characteristics, drug/growth factor delivery and bone regeneration application.” Biomaterials Science, 2012. https://pmc.ncbi.nlm.nih.gov/articles/PMC3363021/
[6] Zhang et al. “A Guided Walk through the World of Mesoporous Bioactive Glasses (MBGs): Fundamentals, Processing, and Applications.” International Journal of Molecular Sciences, 2020. https://pmc.ncbi.nlm.nih.gov/articles/PMC7767440/
[7] Kumar et al. “Reinforcing β-tricalcium phosphate scaffolds for potential applications in bone tissue engineering: impact of functionalized multi-walled carbon nanotubes.” Scientific Reports, 2024. https://www.nature.com/articles/s41598-024-68419-2
[8] Li et al. “Beta-tricalcium phosphate granules improve osteogenesis in vitro and establish innovative osteo-regenerators for bone tissue engineering in vivo.” Scientific Reports, 2016. https://www.nature.com/articles/srep23367
[9] Zara et al. “Porous beta tricalcium phosphate scaffolds used as a BMP-2 delivery system for bone tissue engineering.” Bone, 2010. https://pubmed.ncbi.nlm.nih.gov/19301273/
[10] Kim et al. “Formulation, Delivery and Stability of Bone Morphogenetic Proteins for Effective Bone Regeneration.” Pharmaceutical Research, 2017. https://link.springer.com/article/10.1007/s11095-017-2147-x
[11] Chen et al. “Bone Morphogenetic Protein-2 in Development and Bone Homeostasis.” International Journal of Molecular Sciences, 2020. https://pmc.ncbi.nlm.nih.gov/articles/PMC7557435/
[12] Lee et al. “Current Status of Recombinant Human Bone Morphogenetic Protein-2 (rhBMP-2) in Maxillofacial Surgery: Should It Be Continued?” Biomedicines, 2023. https://www.mdpi.com/2306-5354/10/9/1005
What do you think of this news? Let us know your thoughts! Join the discussion of this and other 3D printing topics at 3DPrintBoard.com.
[Source / Images: ‘A novel vehicle-like drug delivery 3D printing scaffold and its applications for a rat femoral bone repairing in vitro and in vivo’]
Where to Buy
Buy Bone Tissue Engineering Materials on Amazon
Buy Hydroxylapatite 3D Printing Powder on Amazon
📌 Related Articles
- Best 3D Printer Upgrades That Actually Improve Print Quality: Complete 2026 Guide
- Best Budget 3D Printer Upgrades That Actually Improve Print Quality: Belts, Springs, Hotends & More
- 3D Printing Safety Equipment Guide: Respirators, Gloves, and Ventilation for 2026
- Prusa Research Mini+ vs Prusa MK4: Full Specs Comparison & Buyer’s Guide
- Bambu Lab P1S vs Bambu Lab P2S: Full Specs Comparison & Buyer’s Guide