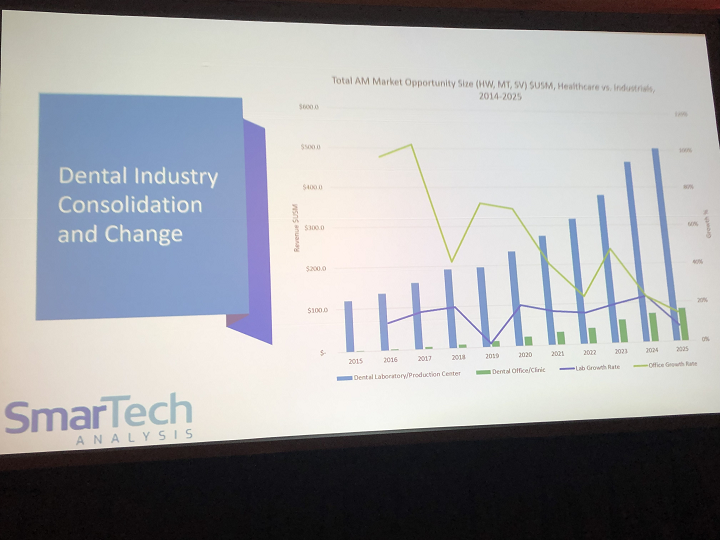
As SmarTech’s VP of Research Scott Dunham said at our recent Additive Manufacturing Strategies event in Boston, the healthcare portion of the AM market is truly the backbone of the industry; combining medical and dental applications, it’s worth a little more than $3 billion dollars. AMS 2020 keynote speaker Dr. Banu Gemici-Ozkan, Senior Market Intelligence Leader for GE Additive, said that dental is the most mature industry for metal AM. In its fifth Additive Manufacturing in Dentistry report, SmarTech notes that “an emerging dental industry transformation is taking place from converging forces,” with 3D printing right at the center.
That’s why LMT Lab Day, which recently concluded in Chicago and is the largest gathering of the dental laboratory community in North America, is seeing more and more 3D printing innovations on the exhibition floor. Over the past week, lots of big announcements have come from the show – Prodways announced the development of its Clear Aligners Manufacturing Ecosystem, BEGO and Formlabs are partnering up for on-demand dental products, EnvisionTEC and dental CAD/CAM software developer exocad are integrating their products for a streamlined digital workflow, etc.
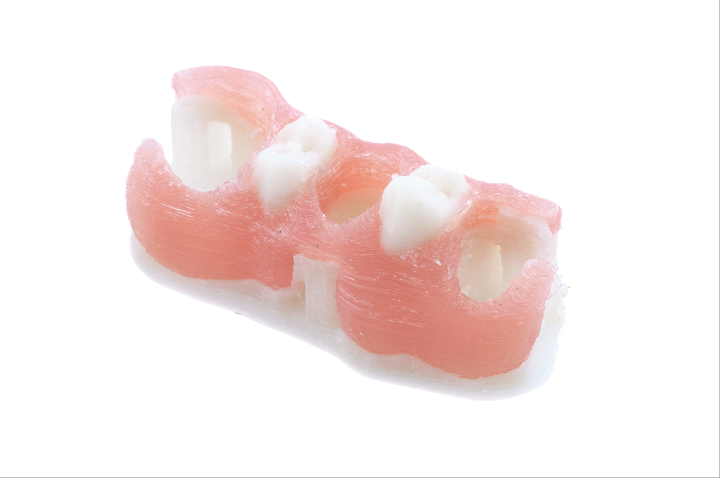
 New York-based dental tech startup Arfona, which was founded in 2016 by several 3D printing enthusiasts and dental technicians, specializes in thermoplastic 3D printed dental prosthetics, and its products are available in over 30 countries around the world. The award-winning startup is actually the inventor of the 3D printed flexible partial denture, and was also at last week’s LMT Lab Day Chicago. Arfona had a few announcements of its own to make – it launched several new products and services at the event.
New York-based dental tech startup Arfona, which was founded in 2016 by several 3D printing enthusiasts and dental technicians, specializes in thermoplastic 3D printed dental prosthetics, and its products are available in over 30 countries around the world. The award-winning startup is actually the inventor of the 3D printed flexible partial denture, and was also at last week’s LMT Lab Day Chicago. Arfona had a few announcements of its own to make – it launched several new products and services at the event.
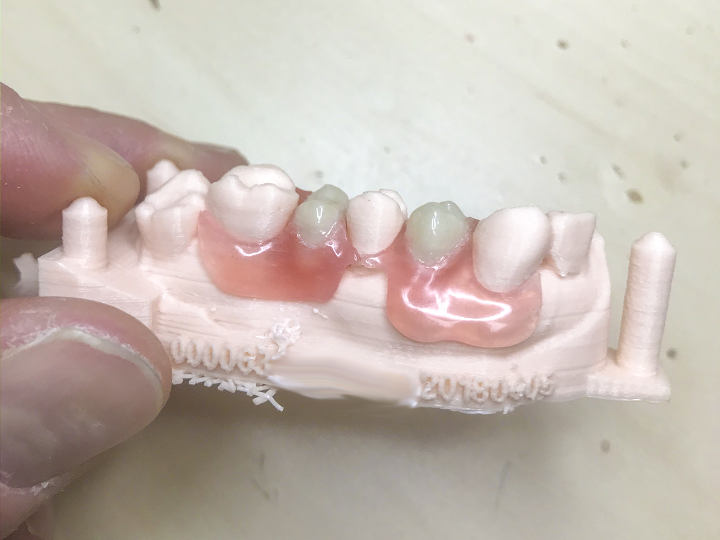
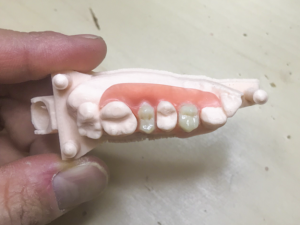 First off, Arfona will be relocating to a new state of the art 3D printing facility in New York. Once the relocation and expansion is complete, the startup’s service bureau department will then offer dental material manufacturer Valplast‘s 3D printing services to dental laboratories. In fact, its new 3D printing center will be completely focused on 3D printed Valplast partial dentures, and will be accepting all intraoral and model scans from dental labs. In addition, Arfona will also be offering 3D print services for custom impression trays, denture try-ins, and low-resolution dental models.
First off, Arfona will be relocating to a new state of the art 3D printing facility in New York. Once the relocation and expansion is complete, the startup’s service bureau department will then offer dental material manufacturer Valplast‘s 3D printing services to dental laboratories. In fact, its new 3D printing center will be completely focused on 3D printed Valplast partial dentures, and will be accepting all intraoral and model scans from dental labs. In addition, Arfona will also be offering 3D print services for custom impression trays, denture try-ins, and low-resolution dental models.
Biocompatible Materials and Processes
The most exciting of Arfona’s announcements is its new FILAdent material – a thermoplastic PMMA filament that can be used to 3D print denture teeth when combined with Valplast’s partial dentures. PMMA, or poly(methyl methacrylate), is inert, biocompatible, and easy to obtain, which is why it’s also been used to make cranial implants.
Arfona’s founder and CEO Justin Marks made the announcement about the startup’s new FILAdent material at LMT Lab Day.
“The combination of FILAdent and Valplast represents the dental industry’s first real use-case for multimaterial and multicolor printing,” stated Marks in a release sent to 3DPrint.com. “Our dual-extrusion r.Pod printer is now capable of producing a Valplast denture base with acrylic teeth in a single build. This revolutionary development for dental 3D printing further reduces the number of steps needed to produce a finished Valplast denture and brings us even closer to our goal of providing affordable tooth replacement for a growing edentulous population.”
Clinical Applications and Case Studies
FILAdent will come in multiple basic tooth shades, and will be made available to users of Arfona’s multimaterial, desktop r.Pod 3D printer in the second half of 2020.
Discuss this story and other 3D printing topics at 3DPrintBoard.com or share your thoughts in the Facebook comments below.
[Images: Arfona, unless otherwise noted]
Regulatory Considerations and Safety
The post Arfona Announces New Dental 3D Printing Products and Services at LMT Lab Day appeared first on 3DPrint.com | The Voice of 3D Printing / Additive Manufacturing.
from Your daily news from 3DPrint.com http://bit.ly/2SYs7SU
Related Articles
Frequently Asked Questions
How is 3D printing used in medicine?
3D printing is used in medicine for surgical planning models, custom implants, bioprinting tissue scaffolds, drug delivery systems, dental aligners, and prosthetics. It enables patient-specific solutions that improve outcomes and reduce surgery time.
What materials are biocompatible for 3D printing?
Common biocompatible materials include PEEK, titanium alloys (Ti6Al4V), bio-ceramics (hydroxyapatite), medical-grade resins, PLA for temporary implants, and hydrogels for bioprinting. Material choice depends on the application and required mechanical properties.
Is 3D printed medical equipment FDA approved?
Yes, several 3D printed medical devices have FDA clearance, including orthopedic implants, dental restorations, and surgical guides. Each device must go through the appropriate regulatory pathway based on its risk classification.
📌 Related Articles
- The Current State of Metal 3D Printing in 2020
- Best 3D Printer Upgrades That Actually Improve Print Quality: Complete 2026 Guide
- Best Budget 3D Printer Upgrades That Actually Improve Print Quality: Belts, Springs, Hotends & More
- 3D Printing News: Multi-Material Breakthrough and Aerospace Market Boom
- 3D Printing Tungsten Carbide: How Hot-Wire Laser Technology Creates Metal Harder Than Steel