“Explore to what extent new materials synthesized through synthetic biology can yield superior structural polymers or resins with programmable lifetimes.”
Bailey Hume, Jesse Rozsa, Hyun Jin Jung, and Mark P. Running are researchers at University of Louisville, focused on a unique area of materials science in bioprinting as detailed in their recently published study, ‘Bioproduction of Molecules for Structural 3D Printing Filaments.’
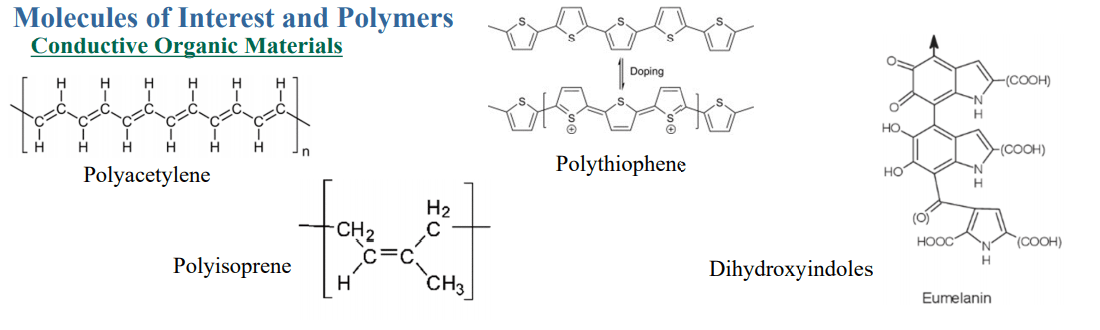
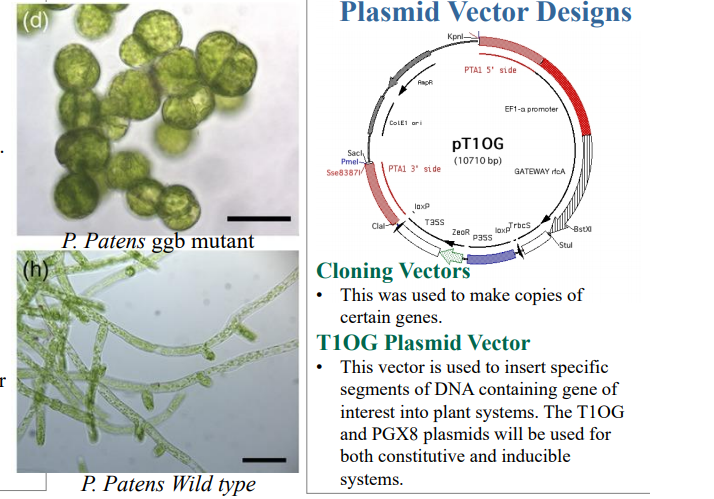
The goal of the research overall was to create a material that is organic, conductive, and biodegradable, resulting from the development of a ggb knockout mutant line of P. patens meant to be ‘immortal’ and offer higher performance then ‘wild-type strains’ used in bioreactors. Affordability was a requirement also as the researchers searched for a renewable source that would also be suitable in an industrial setting, making use of the formidable benefits included with 3D printing.
P. patens is also known as Spreading Earth Moss with a sequenced genome that is often now used in biotechnology in bioreactors and as a model organism.
The Role of 3D Printing in Medicine
The material created through this research is meant to be used by KAMPERS participants,
“The project, titled the Kentucky Advanced Partnership for Enhanced Robotics and Structures (or KAMPERS), will harness the collective research power of 40 multidisciplinary researchers from the eight Kentucky universities and colleges, which include UK, UofL, Eastern Kentucky University (EKU), Kentucky State University (KSU), Morehead State University (MSU), Somerset Community College, Transylvania University (TU) and Western Kentucky University (WKU).”
Biocompatible Materials and Processes
This study is NSF funded and “KAMPERS, will develop materials for flexible electronics, produce 3D printed structures, and integrate sensing and other electronic functions into these structural elements to advance human-machine interfaces. ”
Different model systems were dissected by natural enzymes as the researchers continued on their quest to find natural resources considered to be ‘green’ and able to be broken down organically.
“Common model systems that have been studied include Arabidopsis, tobacco, and maize. DNA is inserted randomly into these model systems in a variety of ways in which insert foreign DNA into a host. Specifically, in Physcomitrella patens homologous recombination gene replacement can be used to insert foreign DNA. This system has been studied extensively over the past decade and we plan to utilize this system to produce green materials for industry,” explained the researchers.
Clinical Applications and Case Studies
Genetic tests/extraction was done through:
- Antibiotic selection
- Electrophoresis
- PCR
- RT-qPCR
- Western blots
- Sequencing
Antibiotics used were meant to create selective cells featuring ‘genes of interest,’ and to be grown in liquid over several days in the lab.
Regulatory Considerations and Safety
The researchers also considered the following:
- Thin layer chromatography – identify of interest materials
- Liquid chromatography – separate materials into fractions
- Gas chromatography – quantify composition of products
To ensure and confirm that genetic modification did, in fact, take place, sequencing was performed to evaluate the targeted genes. Each E.coli cell divides, with vectors also reproducing.
“After the vectors have been harvested from the cells, restriction enzymes will be used to ascertain our gene of interest from the cell,” concluded the researchers. “Finally, these genes of interest will be placed into destination vectors which will undergo transformation into our model system and therefore homologous recombination as well.”
Research Breakthroughs and Innovations
“RNA extraction, agarose gel electrophoresis, and PCR will each be used to evaluate the extent of the genetic modification in the P. patens. To ensure and confirm that genetic modification did, in fact, take place, sequencing will be done to identify the genes of interest in their specific frames.”
What do you think of this news? Let us know your thoughts; join the discussion of this and other 3D printing topics at 3DPrintBoard.com.
[Source / Images: ‘Bioproduction of Molecules for Structural 3D Printing Filaments’]
The post KAMPERS: Bioproduction of Organic, Conductive and Biodegradable 3D Print Materials appeared first on 3DPrint.com | The Voice of 3D Printing / Additive Manufacturing.
Frequently Asked Questions
How is 3D printing used in medicine?
3D printing is used in medicine for surgical planning models, custom implants, bioprinting tissue scaffolds, drug delivery systems, dental aligners, and prosthetics. It enables patient-specific solutions that improve outcomes and reduce surgery time.
What materials are biocompatible for 3D printing?
Common biocompatible materials include PEEK, titanium alloys (Ti6Al4V), bio-ceramics (hydroxyapatite), medical-grade resins, PLA for temporary implants, and hydrogels for bioprinting. Material choice depends on the application and required mechanical properties.
Is 3D printed medical equipment FDA approved?
Yes, several 3D printed medical devices have FDA clearance, including orthopedic implants, dental restorations, and surgical guides. Each device must go through the appropriate regulatory pathway based on its risk classification.
📌 Related Articles