Quick Answer Box: What is Aspiration Assisted Bioprinting?
Aspiration Assisted Bioprinting (AAB) is a revolutionary bioprinting method developed by researchers at Pennsylvania State University that uses suction forces to precisely pick up and place tiny tissue spheroids, cells, and tissue strands into 3D patterns. This cost-effective approach, built on a modified MakerBot Replicator 1, enables unprecedented precision in tissue fabrication for both scaffold-based and scaffold-free bioprinting applications.
In the rapidly evolving fields of tissue engineering and regenerative medicine, an increased interest and use of bioprinting technology is changing the way researchers are conducting investigations. To satisfy particular requirements of their research, many labs around the globe are creating customized bioprinters, developing new bioprinting techniques, and even producing their own bioinks. In the past few years, we have been following news from the Pennsylvania State University (Penn State) biomedical engineering experts as their projects advance bioprinting and fabrication of tissue scaffolding to create living tissue. In their latest development, researchers at Penn State’s College of Engineering have hatched a new method of bioprinting that uses aspiration of tiny biologics – like spheroids, cells, and tissue strands – to precisely place them in 3D patterns, either on scaffolding or without, to create artificial tissues with natural properties.
Understanding Aspiration Assisted Bioprinting (AAB)
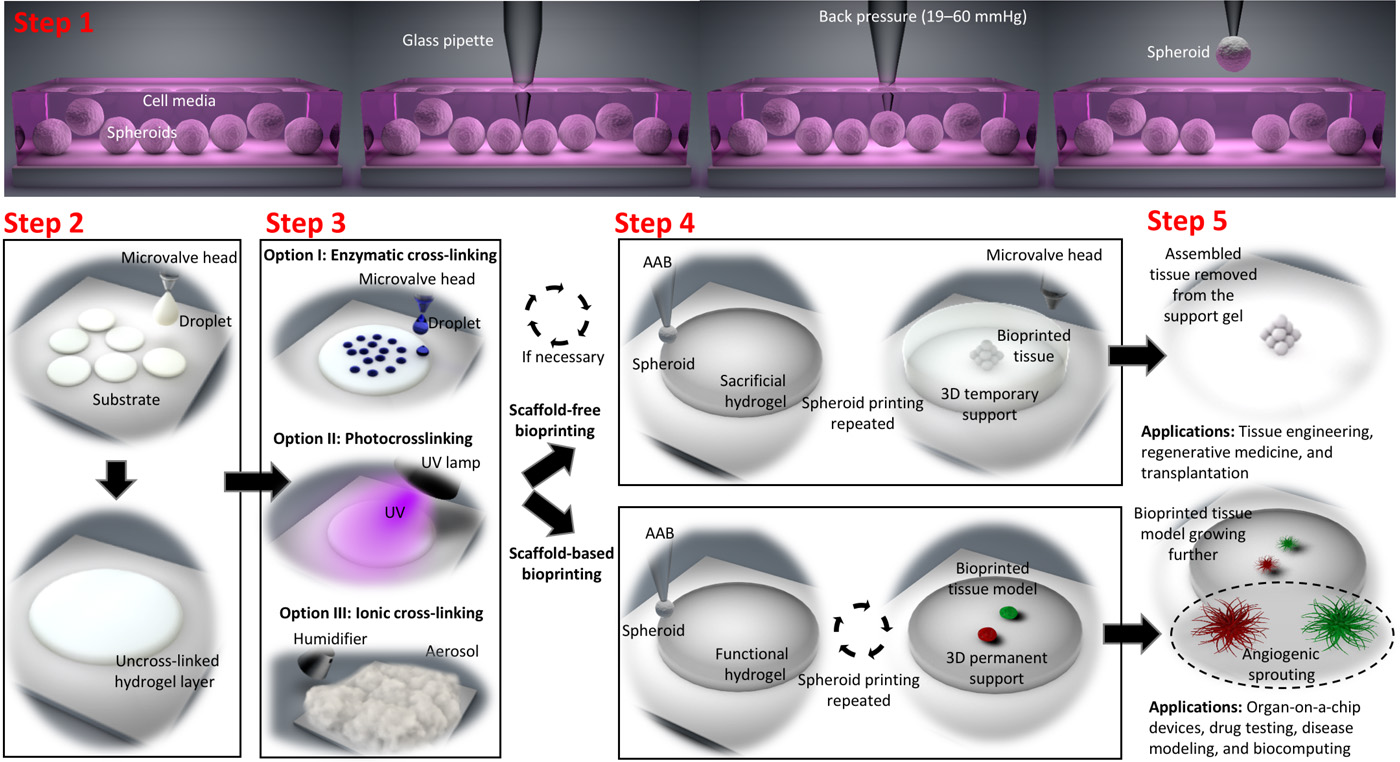
The method, called aspiration-assisted bioprinting (AAB), enables picking and bioprinting biologics in 3D through harnessing the power of aspiration forces. When coupled with microvalve bioprinting, it facilitated different biofabrication schemes, including scaffold-based or scaffold-free bioprinting at an unprecedented placement precision.
Penn State researchers published an article on their tissue engineering work titled: “Aspiration-assisted Bioprinting for Precise Positioning of Biologics,” in the journal Science Advances, whereby Ibrahim T. Özbolat, co-author and Hartz Family Career Development Associate Professor of Engineering Science and Mechanics, and his team used their new AAB method along with conventional micro-valve printing to create homogeneous tissues and tissues containing a variety of cells.
“Tissue spheroids have been increasingly used as building blocks for the fabrication of tissues, but their precise bioprinting has been a major limitation,” said Özbolat. “In addition, these spheroids have been primarily bioprinted in a scaffold-free manner and could not be applied for fabrication with a scaffold.”
The Importance of Scaffolds in Tissue Engineering
Using scaffolding is necessary for many applications in regenerative medicine and tissue engineering and also in the fabrication of microphysiological systems for disease modeling or drug screening. Scaffolds play an important role in creating a 3D environment to induce tissue formation, in fact, the application of scaffolding materials together with stem cell technologies is believed to hold enormous potential for tissue regeneration.
Scaffolds provide structural support for growing cells and help guide tissue development. They can be made from various materials, including natural polymers like collagen and alginate, or synthetic materials like PLGA and PEG. The choice of scaffold material depends on the specific application, degradation rate requirements, and mechanical properties needed for the target tissue.
How Aspiration Assisted Bioprinting Works
According to Penn State, AAB uses the power of suction to move tiny microscopic spheroids. Suggesting that, just as one could pick up a pea by placing a drinking straw on it and sucking through the straw, AAB picks up the tissue spheroid, holds the suction on the spheroid until it is placed in exactly the proper location and then releases it.
“Of course, we have to gently aspirate the spheroids according to their viscoelastic properties so no damage occurs in transferring the spheroids to the gel substrate. The spheroids need to be structurally intact and biologically viable,” Özbolat explained. “We demonstrated for the first time that by controlling the location and distance between spheroids we can mediate collective capillary sprouting.”
The process involves several precise steps:
- Pick-up: A glass pipette applies controlled suction to aspirate a tissue spheroid
- Transport: The spheroid is held securely by the suction force while being moved to the target location
- Placement: The spheroid is precisely positioned using the 3D motion stage
- Release: Suction is released, depositing the spheroid exactly where needed
Creating Complex Tissue Structures
It appears that by controlling the exact placement and type of spheroid, the researchers were able to create samples of heterocellular tissues – those containing different types of cells – as well as create a matrix of spheroids with capillary sprouting in the desired directions. Furthermore, they describe capillaries as necessary for the creation of tissues that can grow and continue to live, so they are a means of delivering oxygen and nutrients to the cells, without which, cells would die. Without capillaries, only the outermost cells will receive oxygen and nutrients.
Penn State specified that the precise placement of spheroids also allows the creation of heterocellular tissues like bone and that by starting their investigation with human mesenchymal stem cells, the researchers found that the cells differentiated and self-assembled bone tissue.
Technical Implementation and Hardware
In the paper, the investigators describe how they modified a MakerBot Replicator 1 (which cost them less than 1000 dollars) to develop the new AAB platform, which operates a custom-made glass pipette that is used to “pick up” biologics and 3D bioprint them into or onto a gel substrate. The extrusion head of the MakerBot was removed and a holder for a pipette and two microvalve heads were 3D printed using an Ultimaker 2 3D printer. The AAB was coupled with microvalves for droplet-based bioprinting of functional or sacrificial hydrogels. And to bioprint spheroids onto a sacrificial hydrogel (such as alginate), microvalve bioprinting and aerosol cross-linking processes were used. To control the 3D motion stage, an Uberlock smoothie board was integrated.
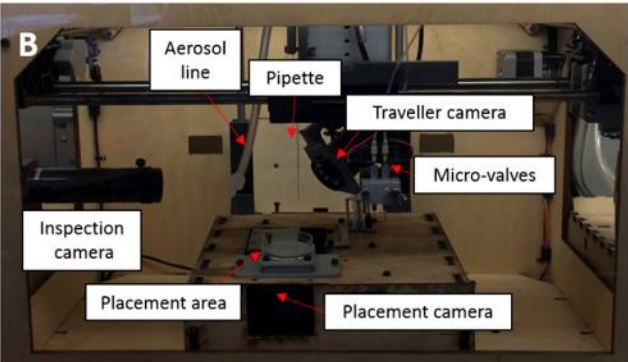
The AAB platform, where micro-valves and a customized pipette head were integrated on the print head (Image: Bugra Ayan, Penn State)
Comparison: AAB vs Traditional Bioprinting Methods
| Feature | Aspiration Assisted Bioprinting | Extrusion Bioprinting | Laser-Assisted Bioprinting | Inkjet Bioprinting |
|---|---|---|---|---|
| Precision | High (micrometer-level) | Medium | Very High | Medium-High |
| Cell Viability | High (gentle handling) | Medium (shear stress) | High | Medium (thermal stress) |
| Cost | Low ($1000+) | Medium | High | Low-Medium |
| Scaffold Support | Yes, both scaffold-based and scaffold-free | Primarily scaffold-based | Both | Primarily scaffold-based |
| Spheroid Handling | Excellent (direct pick-up) | Poor (requires embedding) | Good | Poor |
| Throughput | Medium (manual positioning) | High | Low-Medium | High |
| Material Compatibility | Wide (spheroids, cells, strands) | Limited (hydrogels) | Wide | Limited (low viscosity) |
Applications in Tissue Engineering and Beyond
In addition to spheroids, Özbolat suggested that there may also be other uses for this system. In fact, other living cells and tissue building blocks could benefit from this method, including electrocytes from electric eel and tissue strands that can be bioprinted for a wide variety of applications, such as tissue engineering, regenerative medicine, drug testing and pharmaceutics, disease modeling, microphysiological systems, biophysics, and biocomputing.
Actually, the ability to produce artificial living tissues is valuable in areas outside of regenerative medicine. Frequently, tissue samples are necessary to test drugs or screen other chemical products; other disciplines include: microfluidics, in vitro human disease models, organoid engineering, biofabrication and tissue engineering, biocomputing, and biophysics.
Comparison: Scaffold-Based vs Scaffold-Free Bioprinting
| Aspect | Scaffold-Based Bioprinting | Scaffold-Free Bioprinting | AAB Advantage |
|---|---|---|---|
| Structural Support | Immediate from scaffold material | Self-assembled over time | Can use either approach |
| Vascularization | Requires channels/sacrificial materials | Natural formation possible | Precise spheroid placement aids vascularization |
| Material Integration | Cells embedded in scaffold | Cells only | Can print both spheroids and scaffolds |
| Resolution | Limited by scaffold resolution | Limited by spheroid size | High precision in both modes |
| Mechanical Properties | Determined by scaffold | Self-generated by cells | Can combine both for optimal properties |
| Degradation | Scaffold degrades over time | No foreign material to degrade | Flexible approach based on application |
| Complexity | Higher (multiple materials) | Lower (cells only) | Simplifies complex tissue fabrication |
Organ-on-a-Chip and Microphysiological Systems
This approach is presented as the first bioprinting method that allows high-precision bioprinting of spheroids in both a scaffold-free and a scaffold-based manner. With this custom-made bioprinter, the researchers claimed to be able to exploit it for multiple applications, since they consider that the precise positioning of spheroids can also be critical for applications such as building organ-on-a-chip devices where the proximity of spheroids to each other or a perfusion channel can be crucial for the viability and function of spheroids as well as the robustness of the developed system.
The paper proposes that “organoids or spheroids are currently loaded using manual approaches, which may reduce the repeatability of system outcome measurements (such as measured insulin in circulating perfusion media in a pancreas-on-a-chip model).” While AAB proposes “a more effective strategy, taking advantage of a simple-to-use, cost-effective and reproducible tissue bioprinting platform.”
Organ-on-a-chip systems are microfluidic cell culture devices that simulate organ-level physiology. These systems are increasingly important for drug development, toxicology testing, and personalized medicine. The ability to precisely position different cell types and spheroids within these chips could dramatically improve their physiological relevance and predictive power.
Future Development and Challenges
According to the researchers of the project, which was supported by the National Science Foundation, the National Institutes of Health and Penn State’s Materials Research Institute, the system still needs improvement to print spheroids in high-throughput to create larger tissues in a shorter time. Actually, scalable tissue fabrication is one of the most important aspects of many bioprinting research initiatives. To create scalable, structurally-stable tissue constructs in the future, customized bioprinting methods like this one can help to eliminate certain roadblocks in the field.
Key challenges for future development include:
- Throughput Scaling: Increasing the speed of spheroid placement to fabricate larger tissues
- Automation: Developing automated spheroid loading and positioning systems
- Multi-Material Integration: Combining different spheroid types and scaffold materials seamlessly
- Vascular Network Formation: Enhancing capillary sprouting for thicker tissue constructs
- Long-Term Viability: Ensuring bioprinted tissues remain functional over extended periods
There are some remarkable advances in bioprinting and we are ecstatic to hear more about them, as well as the challenges that researchers are facing to make this new field one of the most relevant and necessary to advance ideal medical treatments as well as so many other disciplines.
Frequently Asked Questions (FAQ)
Q: What makes Aspiration Assisted Bioprinting different from other bioprinting methods?
A: AAB is unique because it uses suction forces to directly pick up and place tissue spheroids with high precision, unlike extrusion methods that require cells to be embedded in bioinks. This gentler handling maintains higher cell viability and allows both scaffold-based and scaffold-free printing approaches.
Q: How much does it cost to build an AAB system?
A: The Penn State team built their AAB platform by modifying a MakerBot Replicator 1 (under $1000), adding a custom glass pipette, microvalves, and 3D-printed holders. This makes it significantly more accessible than high-end commercial bioprinters that can cost tens or hundreds of thousands of dollars.
Q: What types of tissues can be created using AAB?
A: AAB has been demonstrated to create various tissue types including heterocellular tissues like bone, tissues with capillary networks, and structures containing multiple cell types. It’s particularly suited for organ-on-a-chip applications and microphysiological systems where precise cell positioning is critical.
Q: Can AAB print complete organs for transplantation?
A: While AAB is a significant advance, it’s not yet capable of printing complete, transplant-ready organs. The current system is ideal for creating tissue models for research, drug testing, and disease modeling. Scaling up to full organs will require further development in throughput, vascularization, and long-term tissue viability.
Q: What are the main advantages of AAB for researchers?
A: The main advantages include: (1) High precision placement of spheroids, (2) Low cost and DIY-friendly design, (3) Gentle handling that preserves cell viability, (4) Flexibility to work with both scaffold-based and scaffold-free approaches, and (5) Reproducibility compared to manual spheroid placement methods.
Q: How long did it take the Penn State team to develop this system?
A: The development process involved designing and fabricating custom components, modifying the MakerBot platform, and extensive testing with various tissue types. While the exact timeline isn’t specified in the paper, this represents years of research in Özbolat’s laboratory focused on advancing bioprinting technologies.
Q: Is the AAB system available for other labs to use?
A: While the specific hardware may not be commercially available, the researchers published detailed specifications and methods in Science Advances. The open-source nature of the components (MakerBot, custom 3D-printed parts) means other labs could potentially replicate or adapt this system for their own research needs.
Conclusion
Aspiration Assisted Bioprinting represents a significant step forward in making precise, accessible bioprinting technology available to researchers worldwide. By combining low-cost components with innovative suction-based handling of tissue spheroids, the Penn State team has created a versatile platform that can advance tissue engineering, drug testing, and organ-on-a-chip research. While challenges remain in scaling the technology for larger tissue constructs, the foundation laid by AAB opens new possibilities for researchers working to solve some of medicine’s most pressing challenges.
The future of bioprinting depends on innovations that balance precision, cost-effectiveness, and biological fidelity. AAB demonstrates that sometimes the most impactful solutions come from reimagining how we manipulate biological materials at the microscale. As this technology continues to evolve, it may help accelerate the development of life-saving therapies, reduce animal testing through better in vitro models, and ultimately bring us closer to the goal of engineering replacement tissues and organs.
Related: AI Driven Bioprinting Speeds up Tissue Engineering · Startup Accelerator: Volumetric Aims to Be the Tissue Bioprinting Farm of the Fu · The Progress & Ongoing Challenge of 3D Bioprinting Cardiac Tissue
Frequently Asked Questions
How is 3D printing used in medicine?
3D printing is used in medicine for surgical planning models, custom implants, bioprinting tissue scaffolds, drug delivery systems, dental aligners, and prosthetics. It enables patient-specific solutions that improve outcomes and reduce surgery time.
What materials are biocompatible for 3D printing?
Common biocompatible materials include PEEK, titanium alloys (Ti6Al4V), bio-ceramics (hydroxyapatite), medical-grade resins, PLA for temporary implants, and hydrogels for bioprinting. Material choice depends on the application and required mechanical properties.
Is 3D printed medical equipment FDA approved?
Yes, several 3D printed medical devices have FDA clearance, including orthopedic implants, dental restorations, and surgical guides. Each device must go through the appropriate regulatory pathway based on its risk classification.
📌 Related Articles
- Best 3D Printer Upgrades That Actually Improve Print Quality: Complete 2026 Guide
- Best Budget 3D Printer Upgrades That Actually Improve Print Quality: Belts, Springs, Hotends & More
- Prusa Research Mini+ vs Prusa MK4: Full Specs Comparison & Buyer’s Guide
- Bambu Lab A1 vs Elegoo Centauri Carbon: Full Specs Comparison & Buyer’s Guide
- ABS 3D Printing Settings Guide: Temperature, Enclosure, and Cooling for Strong Parts