Researchers from Germany and China are working together to study the use of hydrogels in bioprinting further, outlining their findings in the recently published ‘3D printing of hydrogels: Rational design strategies and emerging biomedical applications.’
Because hydrogels are deeply linked to bioprinting, they are the focus of many different research projects around the world, from studying impacts on conductivity to adding using hydrogels in direct ink writing, fabricating them in ongoing new techniques, and more. In this study, the scientists focus on current designs, techniques, material, and properties related to successful use with hydrogels—as well as reminding us that ‘3D printing is considered as the next global industrial and manufacturing revolution.’
Aside from bioprinting and tissue engineering, the applications in which 3D printing can be used are endless, from automotive to chemical, medical, pharmaceutical, robotics, energy…and the list continues go on.
“To fully unleash the huge potential of 3D printing, new printable inks made of different materials are needed, and this requires on-demand design and development of the composition, structure, function, and dynamics of the ink materials,” state the authors.
The Role of 3D Printing in Medicine
And while 3D printing continues to transform tissue engineering, offering control, speed, accuracy, and often great affordability too, the need for better inks still constricts many research projects. With the use of hydrogel networks, however, researchers can manipulate ‘matrix remodeling,’ cell migration and sustainability, and adhesion too. These structures can be customized in terms of material, chemistry, and properties. Because they can mimic the extracellular matrix (ECM) formidably, hydrogels are able to ‘direct cell fate’ when used as 3D printable inks.
“From the standpoint of tissue engineering, establishing a cell microenvironment that can mimic the native ECM is highly desired and meaningful to recapitulate in vivo milieu and replicate cell/tissue functions in vitro,” explain the researchers. “By mimicking ECM, hydrogels are capable of not only supplying structural support for cell residence but also affording various predefined biochemical (cytokines, growth factors, cell adhesion peptides, etc.) and biophysical (structures, stiffness, degradation, etc.) cues for modulating cell fate.”
Biocompatible Materials and Processes
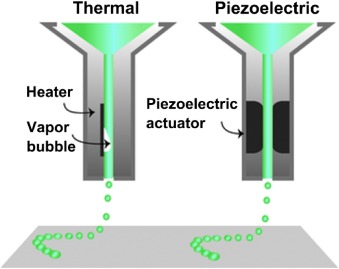
In bioprinting, there are several different 3D printing methods, to include laser-based systems, nozzle-based systems, and printer-based systems.

Clinical Applications and Case Studies
Schematic diagram to illustrate the working principles of extrusion-based 3D printing technique.
“Nevertheless, not all the 3D printing techniques are applicable to process hydrogel materials since the processing of hydrogel materials needs mild conditions rather than harsh conditions and therefore, it is not the objective of this review to discuss all of these 3D printing techniques,” stated the authors.
Regulatory Considerations and Safety
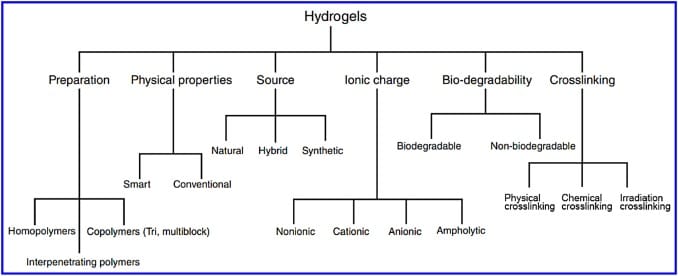
The types of hydrogels can be separated into:
- Natural polymer hydrogels
- Polysaccharide hydrogels
- Polyacrylate hydrogels
- Nanocomposite hydrogels
- Composite hydrogels
- Supramolecular hydrogels
- Stimuli-responsive hydrogels
Research Breakthroughs and Innovations
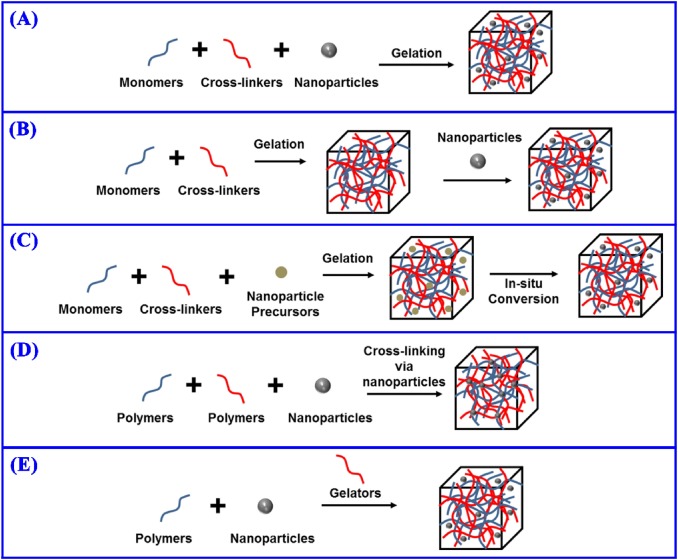
Five major ways for the fabrication of hydrogel–nanoparticle composites with uniform distribution. (A) Synthesizing hydrogels in the suspension of nanoparticles. (B) Physically incorporating nanoparticles into the hydrogel matrices after gelation. (C) In situ synthesis of nanoparticles from precursors embedded in the hydrogel matrices after gelation. (D) Using the nanoparticles as cross-linker to generate hydrogels. (E) Fabricating the hydrogels using polymers, nanoparticles and gelators.
Scaffold designs must meet several different requirements:
- They must be highly porous
- Materials must be biocompatible
- Surfaces must be suitable for cell viability
- Must be able to support required mechanical properties
For suitability in fabrication, hydrogels must possess the correct rheological properties, as well as cross-linking mechanisms.
The Future of Bioprinting and Medical AM
Bio-inks must also be chosen very carefully, with options such as
- Single component hydrogel ink
- IPN hydrogel inks
- Supramolecular hydrogel inks
- Nanocomposite hydrogel inks
The Role of 3D Printing in Medicine
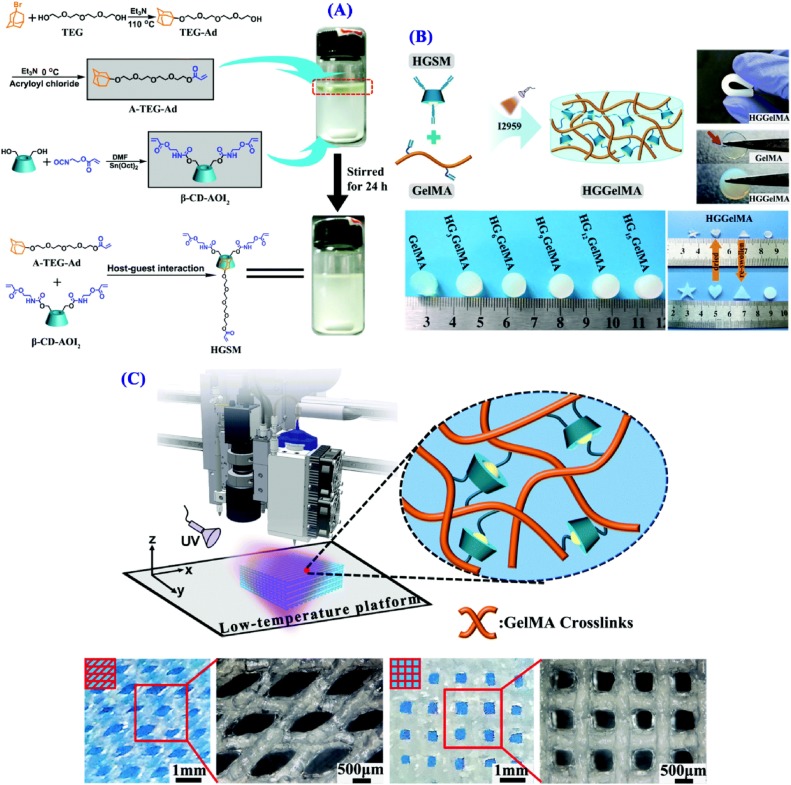
3D printing of self-healing GelMA hydrogels with host–guest interactions. (A) Schematic of HGSM synthesis. (B) Photos of HGGelMA and GelMA hydrogel samples. (C) 3D printing of HGGelMA hydrogel scaffolds.
Bioprinting offers enormous advantages in tissue modeling, allowing for precision in tailoring cell placement, ECM, materials, growth factors in scaffolds, and more. See also: Best Budget 3D Printer Upgrades That Actually Impr…. Tissue-on-a-chip and organ-on-a-chip platforms are major sources of interest too for researchers seeking to imitate the human tissue/organ microenvironment.
“The combination of microfluidics and 3D printing technologies in tissue-on-a-chip and organ-on-a-chip enables more powerful designs for creating complicated flow channels/chambers and functional bioconstructs with 3D heterogeneous structure, cell placement and tissue specificity to recapitulate and reproduce the natural tissue/organ features,” state the authors.
Biocompatible Materials and Processes
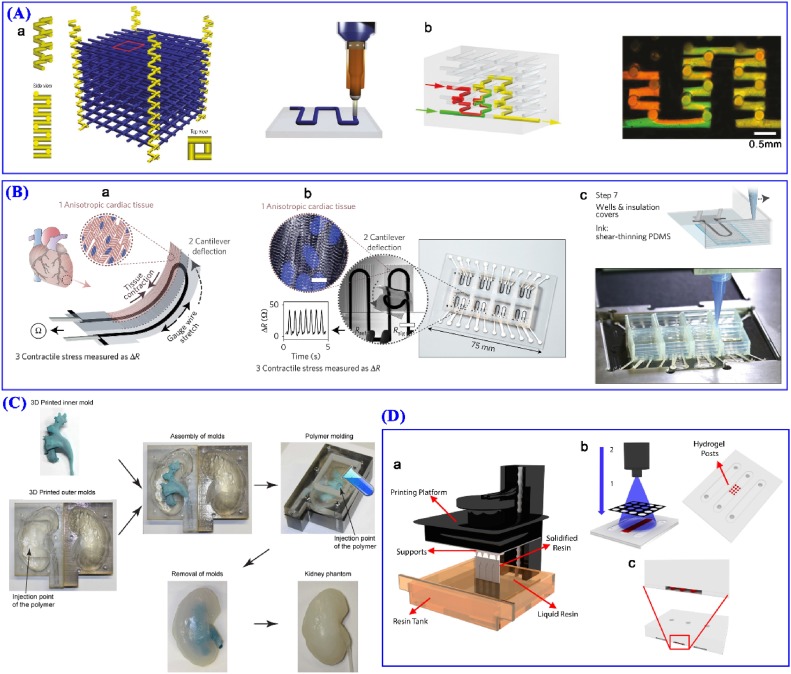
3D printing for microfluidics. (A) 3D microvascular networks fabrication. (a) Schematic of fabricating microvascular scaffold. (b) Microfluidic mixing test. Reproduced with permission [1727]. Copyright 2003, Nature Publishing Group. (B) Multimaterial 3D printing of cardiac microphysiological device. (a) Device principle schematic. (b) Fully printed final device. (c) Microscale 3D printing process (step 7 shown here). Reproduced with permission [1728]. Copyright 2017, Nature Publishing Group. (C) 3D printing of human kidney phantom. Reproduced with permission [1729]. Copyright 2016, The Authors. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://bit.ly/3d1YBnl), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. (D) 3D printing of microfluidic chip. (a) Stereolithography printing process. (b) Hydrogel post formation. (c) 3D chip cross-section. Reproduced with permission [1730]. Copyright 2016, IOP Publishing Ltd.
4D printing and 4D bioprinting are entering the picture more often too as researchers realize a need and see the efficacy of creating smart materials able to adapt to their environments. They can morph or ‘deform’ from their original shape as necessary (often due to moisture or temperature change), and then revert.
“All in all, someday the 3D bioprinting scenes of an organ or a human in science fiction movies will no longer be science fiction but become reality, entering our real world.”
Clinical Applications and Case Studies
What do you think of this news? Let us know your thoughts! Join the discussion of this and other 3D printing topics at 3DPrintBoard.com.
[Source / Images: ‘3D printing of hydrogels: Rational design strategies and emerging biomedical applications’]
The post Hydrogels in Bioprinting Today: Design Strategies for Emerging Applications appeared first on 3DPrint.com | The Voice of 3D Printing / Additive Manufacturing.
from Your daily news from 3DPrint.com http://bit.ly/3b03mfu
Related Articles
Frequently Asked Questions
How is 3D printing used in medicine?
3D printing is used in medicine for surgical planning models, custom implants, bioprinting tissue scaffolds, drug delivery systems, dental aligners, and prosthetics. It enables patient-specific solutions that improve outcomes and reduce surgery time.
What materials are biocompatible for 3D printing?
Common biocompatible materials include PEEK, titanium alloys (Ti6Al4V), bio-ceramics (hydroxyapatite), medical-grade resins, PLA for temporary implants, and hydrogels for bioprinting. Material choice depends on the application and required mechanical properties.
Is 3D printed medical equipment FDA approved?
Yes, several 3D printed medical devices have FDA clearance, including orthopedic implants, dental restorations, and surgical guides. Each device must go through the appropriate regulatory pathway based on its risk classification.
📌 Related Articles
- The Current State of Metal 3D Printing in 2020
- Best Budget 3D Printer Upgrades That Actually Improve Print Quality: Belts, Springs, Hotends & More
- How Divergent Technologies Is 3D Printing Cruise Missiles for the U.S. Military
- The Ultimate Guide to 3D Printer Calibration: Complete Step-by-Step Manual
- Best 3D Printer Upgrades That Actually Improve Print Quality: Complete 2026 Guide