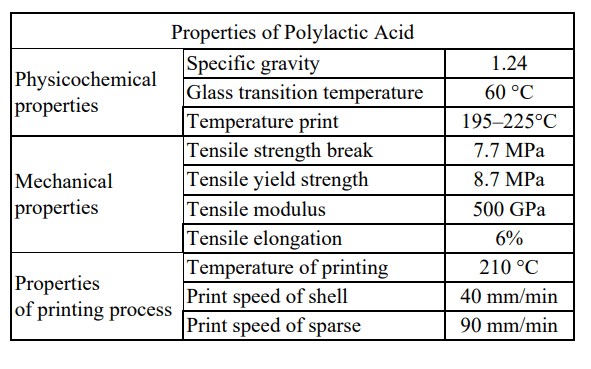
Researchers from Slovakia are delving further into the uses of PLA in bioprinting, releasing their findings in the recently published, ‘3D printed Polylactid Acid based porous scaffold for bone tissue engineering: an in vitro study.’
The team created samples in the form of PLA scaffolds, assessing for both cytotoxicity and biocompatibility, with the ultimate goal of bone tissue engineering meant to assist in bone regeneration—one of the most challenging areas. Researchers have performed a wide range of different studies regarding bioprinting and bone regeneration, creating a variety of structures, scaffolds, and using different printing methods.
PLA has been used before, commonly, and while here the researchers use a commercially available type of PLA scaffolds, they separated samples into three different groups, each pre-treated with:
- Complete culture medium
- Bovine fetal serum
- Human blood
Further, the research team analyzed periosteum-derived cells in terms of cytotoxicity and biocompatibility.
Scaffolds were created using FFF 3D printing (bq Witbox), with scaffold samples of 10 ´ 10 ´ 4 mm and porosity of 61%. Periosteum was taken from the proximal tibia of a 55-year-old female patient who was undergoing a knee replacement surgery. Her consent was given and the Ethical Committee of The University Hospital of Louis Pasteur in Košice approved the process.
“Subsequently, cells were collected by centrifugation at 150 ´ g for 7 min and seeded 25,000 cells/cm2 in a 25 cm2 culture flask (T25) containing Alpha-modified Minimal Essential Medium (Invitrogen, GIBCO®, USA) supplemented with 10% foetal bovine serum (FBS, Invitrogen, GIBCO®, USA) and 1% ATB,” explained the researchers.
“Non-adherent cells were removed after 5 days by changing the medium. Adherent cells were cultured under standard culture conditions at 37 °C in 5% CO2 humidified atmosphere and the medium was replaced every 2–3 days. Confluent cell layers were dissociated with 0.05% Trypsin-EDTA solution (Invitrogen, GIBCO®, USA) and the number and viability of cells was assessed by TC10
Automated Cell Counter (Bio-Rad Laboratories). Periosteum – derived osteoprogenitor cells (PDO) from the third passage (P3) – were used for the flow cytometry analysis and co-cultivation with scaffold.”
The scaffolds were sterilized, and then separated into the three groups:
“First group was incubated within human serum, second in complete medium containing Alpha- -modified minimal essential medium supplemented with 10% FBS and 1% ATB, and third in 10% FBS.”

Grafical outputs of scaffolds-internal structure of scaffold:
A) Top View (119%), B) Right view (167%)
Cells were measured four times and compared to the control group. The results yielded good biocompatibility; however, the researchers noted the best results in the scaffolds coated with human serum. This treatment also encouraged cell growth.
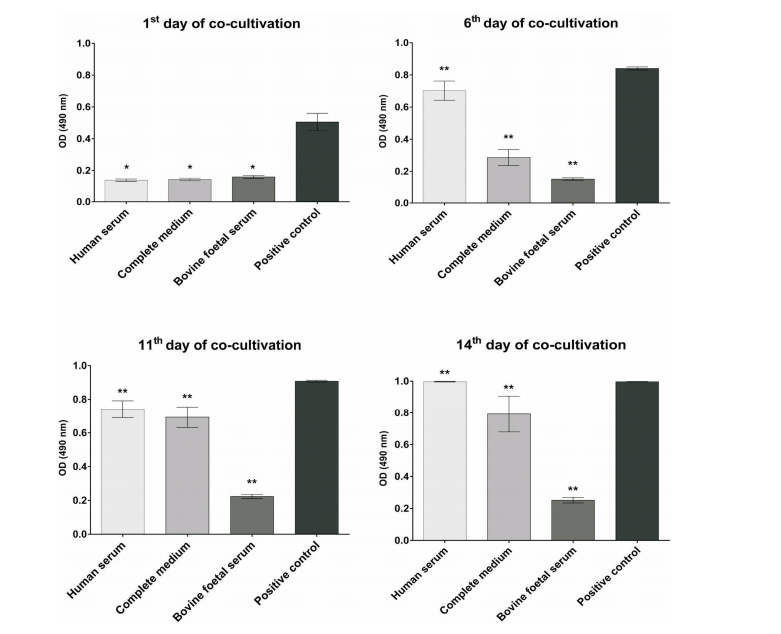
Proliferation of periosteum derived osteoprogenitors during co-cultivation with PLA scaffolds, measured after 1, 6, 11, and 14 days, respectively by CellTiter 96® AQueous One Solution Cell Proliferation Assay. See also: Best 3D Printer Upgrades That Actually Improve Pri…. Data represent mean ± SD value of four independent measures and value of p was p < 0.05 the first day of co-cultivation (*) or p < 0.01 in other days of co-culture (**)
“The distribution, adhesion and proliferation of human PDO on the native PLA scaffolds were also examined using SEM observation during two weeks of cultivation. The human PDOs showed good viability in the scaffolds, which were incubated for 48 hours in human serum, which was expressed by enhanced cellular spreading and proliferation and the pH of media, in which scaffolds and cells were co-cultivated, was 7.4 after 14 days. The pH reached the value of the one in human blood,” concluded the researchers.
“The obtained PLA porous scaffolds favored attachment periosteum derived progenitors and proliferation, furthermore, cells penetrated into the scaffold through the interstitial pores, which was meaningful for cytocompatibility evaluation. New strategies, such as poly-therapy by using scaffolds, healing promotion factors and stem cells, and finally three-dimensional printings, are in their preliminary stages, but may offer new exciting alternatives in the near future.”
What do you think of this news? Let us know your thoughts! Join the discussion of this and other 3D printing topics at 3DPrintBoard.com.
[Source / Images: ‘3D printed Polylactid Acid based porous scaffold for bone tissue engineering: an in vitro study’]
The post Slovakia: Experimenting with 3D Printed PLA Scaffolds for Bone Regeneration appeared first on 3DPrint.com | The Voice of 3D Printing / Additive Manufacturing.
from Your daily news from 3DPrint.com http://bit.ly/2IGeZMb
Where to Buy Filament
Disclosure: Some of the links below are affiliate links. If you make a purchase, we may earn a commission at no extra cost to you. This helps support this site.
- PLA: Hatchbox
Looking for Filament? Check the best deals on Amazon:
These links go to Amazon with our affiliate tag. If you prefer to support local retailers or buy directly from manufacturers, we encourage that as well!
Related Articles
Frequently Asked Questions
How is 3D printing used in medicine?
3D printing is used in medicine for surgical planning models, custom implants, bioprinting tissue scaffolds, drug delivery systems, dental aligners, and prosthetics. It enables patient-specific solutions that improve outcomes and reduce surgery time.
What materials are biocompatible for 3D printing?
Common biocompatible materials include PEEK, titanium alloys (Ti6Al4V), bio-ceramics (hydroxyapatite), medical-grade resins, PLA for temporary implants, and hydrogels for bioprinting. Material choice depends on the application and required mechanical properties.
Is 3D printed medical equipment FDA approved?
Yes, several 3D printed medical devices have FDA clearance, including orthopedic implants, dental restorations, and surgical guides. Each device must go through the appropriate regulatory pathway based on its risk classification.
📌 Related Articles
- The Complete 3D Printing Filament Guide: Types, Properties & When to Use Each
- Best 3D Printer Upgrades That Actually Improve Print Quality: Complete 2026 Guide
- Best Budget 3D Printer Upgrades That Actually Improve Print Quality: Belts, Springs, Hotends & More
- The Ultimate Guide to 3D Printer Calibration: Complete Step-by-Step Manual
- Best 3D Printers Under $500: 2026 Comparison Guide